|
受注:045-509-1970 |
技術サポート:[email protected] 平日9:00〜18:00 1営業日以内にご連絡を差し上げます |
化学情報
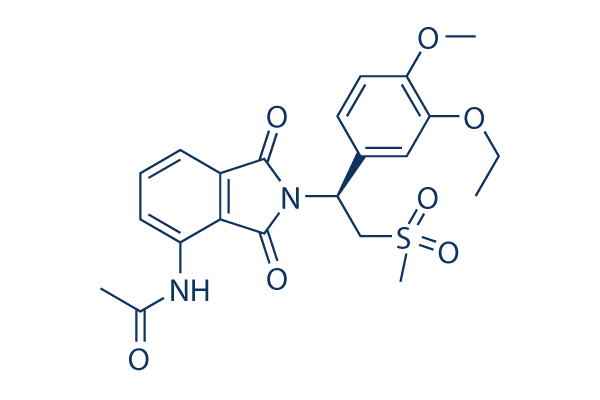
|
Synonyms | CC-10004 | Storage (From the date of receipt) |
3 years -20°C powder 1 years -80°C in solvent |
| 化学式 | C22H24N2O7S |
|||
| 分子量 | 460.5 | CAS No. | 608141-41-9 | |
| Solubility (25°C)* | 体外 | DMSO | 92 mg/mL (199.78 mM) | |
| Water | Insoluble | |||
| Ethanol | Insoluble | |||
|
* <1 mg/ml means slightly soluble or insoluble. * Please note that Selleck tests the solubility of all compounds in-house, and the actual solubility may differ slightly from published values. This is normal and is due to slight batch-to-batch variations. |
||||
溶剤液(一定の濃度)を調合する
生物活性
| 製品説明 | Apremilast (CC-10004) is a potent and orally active PDE4 and TNF-α inhibitor with IC50 of 74 nM and 77 nM, respectively. |
|---|---|
| in vitro | Apremilast is more potent for inhibition of PDE4 compared with cAMP or cGMP hydrolysing enzymes from other PDE families. Apremilast displays a broad pattern of anti-inflammatory activity in a variety of cell types, inhibits TNF-α, IL-12 and IL-23 production, as well as NK and keratinocyte responses. Apremilast is found to inhibit the zymosan-induced PMN production of IL-8 with IC50 of 94 nM. Apremilast inhibits fMLF-induced PMN CD18 and CD11b expression with IC50 of 390 nM and 74 nM, respectively, and inhibits fMLF-induced adhesion of PMN to HUVECs with IC50 of 150 nM. Apremilast inhibits keratinocyte TNF-αproduction, with no effect on keratinocyte cell viability as measured by intracellular ATP levels. [3] [4]. |
| in vivo | Apremilast is stable in the presence of human microsomes (t1/2 > 60 min). It is 90% protein bound in human plasma. Oral and intravenous administration of it in female rats showed that it have good pharmacokinetics with low clearance, a moderate volume of distribution, and a 64% oral bioavailability. In a LPS-induced TNF-αinhibition model in rats, examined the TNF-α inhibitory ability of Apremilast in vivo, and the ED50 is determined to be 0.03 mg/kg. In another LPS-induced neutrophilia model in rats, Apremilast exhibited an ED50 range from 0.3 mg/kg to 0.9 mg/kg.[1] |
プロトコル(参考用のみ)
| キナーゼアッセイ | PDE4 inhibitory activity | |
|---|---|---|
| Cells (1×109) are washed in PBS and lysed in cold homogenization buffer (20 mM Tris-HCl, pH 7.1, 3 mM 2-mercaptoethanol, 1 mM MgCl2, 0.1 mM EGTA, 1 μM PMSF, 1 μg/mL leupeptin). Following homogenization in a Dounce homogenizer the supematant is collected by centrifugation and loaded onto a Sephacryl S-200 column equilibrated in homogenization buffer. PDE is eluted in homogenization buffer and the rolipram sensitive fractions pooled and stored in aliquots. Enzyme activity is assayed in 50 mM Tris- HCl, pH 7.5, 5 mM MgCl2 and 1 μM cAMP (of which 1% is3H cAMP) in the presence of varying concentrations of inhibitors. The amount of extract used is pre-determined to ensure that reactions are within the linear range and consumed lessthan15%of the total substrate. Reactions are performed at 30°C for 30 min and terminated by boiling for 2 min. The samples are then chilled and treated with snake 'venom (1mg/mL) at 30 °C for 15 min. Unused substrate is removed by incubation with 200 μL AG1-X8 resin for 15 min. Samples are then spun at 3000 rpm for 5 min and 50 μL of the aqueous phase taken for counting. Eachdata point is carried out in duplicate with activity expressed as percentage of control. IC50 is determined from dose response curves derived from three independent experiments. | ||
| 細胞アッセイ | 細胞株 | HEKn cells |
| 濃度 | ~10 μM | |
| 反応時間 | 18 h | |
| 実験の流れ | For proliferation studies, human neonatal foreskin epidermal keratinocytes (HEKn cells) are plated at 3000 cells per well in 96-well flat bottom tissue culture plates for 2 days. HEKn cells are treated with apremilast or 0.1% DMSO as the vehicle control for 1 h before ultraviolet B (UVB) irradiation with 50 mJ/cm2 in a UV Stratalinker 2400, calibrated with 312 nm UVB bulbs. Media and compounds are replaced, and cells are incubated for 18 h. Lysates are transferred to plates and shaken for 2 min before chemiluminescence is read on a TopCount NXT Luminescence Counter. | |
| 動物実験 | 動物モデル | Psoriasis mouse model |
| 投薬量 | 5 mg/kg/day, divided into b.i.d.doses | |
| 投与方法 | Oral | |
参考
Selleckの高級品が、幾つかの出版された研究調査結果(以下を含む)で使われた:
| Intestinal dysbiosis exacerbates the pathogenesis of psoriasis-like phenotype through changes in fatty acid metabolism [ Signal Transduct Target Ther, 2023, 8(1):40] | PubMed: 36710269 |
| Targeting PDE4 as a promising therapeutic strategy in chronic ulcerative colitis through modulating mucosal homeostasis [ Acta Pharm Sin B, 2022, 12(1):228-245] | PubMed: 35127382 |
| Phosphodiesterase-4 Inhibition Reduces Cutaneous Inflammation and IL-1β Expression in a Psoriasiform Mouse Model but Does Not Inhibit Inflammasome Activation [ Int J Mol Sci, 2021, 22(23)12878] | PubMed: 34884681 |
| Application of a newly-developed cynomolgus macaque BiTE-mediated cytotoxic T-lymphocyte activity assay to various immunomodulatory agents in vitro [ J Immunotoxicol, 2021, 18(1):154-162] | PubMed: 34714999 |
| Targeting PDE4 as a promising therapeutic strategy in chronic ulcerative colitis through modulating mucosal homeostasis [ Acta Pharmaceutica Sinica B, 2021, 10.1016/j.apsb.2021.04.007] | PubMed: None |
| Apremilast Regulates the Teff/Treg Balance to Ameliorate Uveitis via PI3K/AKT/FoxO1 Signaling Pathway [ Front Immunol, 2020, 11:581673] | PubMed: 33281814 |
| Inhibition of phosphodiesterase-4 attenuates murine ulcerative colitis through interference with mucosal immunity [ Br J Pharmacol, 2019, 176(13):2209-2226] | PubMed: 30883697 |
| Correlation and cluster analysis of immunomodulatory drugs based on cytokine profiles [Wallner FK Pharmacol Res, 2018, 128:244-251] | PubMed: 29079427 |
| Evaluation of clustering methods for analyzing drug cytokine profiles [ DiVA, 2017, None] | PubMed: None |
長期の保管のために-20°Cの下で製品を保ってください。
人間や獣医の診断であるか治療的な使用のためにでない。
各々の製品のための特定の保管と取扱い情報は、製品データシートの上で示されます。大部分のSelleck製品は、推薦された状況の下で安定です。製品は、推薦された保管温度と異なる温度で、時々出荷されます。長期の保管のために必要とされてそれと異なる温度で、多くの製品は、短期もので安定です。品質を維持するが、夜通しの積荷のために最も経済的な貯蔵状況を用いてあなたの送料を保存する状況の下に、製品が出荷されることを、我々は確実とします。製品の受領と同時に、製品データシートの上で貯蔵推薦に従ってください。
