|
受注:045-509-1970 |
技術サポート:[email protected] 平日9:00〜18:00 1営業日以内にご連絡を差し上げます |
化学情報
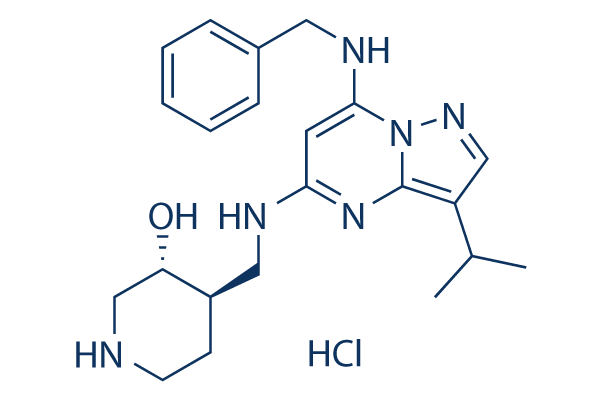
|
Synonyms | CT7001 hydrochloride | Storage (From the date of receipt) |
3 years -20°C powder | |||||||
| 化学式 | C22H31ClN6O |
||||||||||
| 分子量 | 430.97 | CAS No. | 1805789-54-1 | ||||||||
| Solubility (25°C)* | 体外 | DMSO | 86 mg/mL (199.54 mM) | ||||||||
| Water | 86 mg/mL (199.54 mM) | ||||||||||
| Ethanol | 15 mg/mL (34.8 mM) | ||||||||||
| 体内 (毎回新しく調製した物を用意してください) |
|
||||||||||
|
* <1 mg/ml means slightly soluble or insoluble. * Please note that Selleck tests the solubility of all compounds in-house, and the actual solubility may differ slightly from published values. This is normal and is due to slight batch-to-batch variations. |
|||||||||||
溶剤液(一定の濃度)を調合する
生物活性
| 製品説明 | Samuraciclib (ICEC0942) hydrochloride is a new, orally bioavailable CDK7 inhibitor with an IC50 of 40nM. The IC50 values for CDK1, CDK2, CDK5 and CDK9 were 45-, 15-, 230- and 30-fold higher. ICEC0942 (CT7001) promotes cell cycle arrest and apoptosis. |
|---|---|
| in vitro | A wide range of cancer types are sensitive to CDK7 inhibition by ICEC0942 with GI50 values ranging between 0.2-0.3 µM. ICEC0942 inhibits PolII, CDK1, CDK2 and RB (retinoblastoma) phosphorylation in the MCF7 breast cancer cell line in a time and dose-sependent manner. ICEC0942 inhibits phosphorylation of CDK7 substrates and promotes cell cycle arrest and apoptosis[1]. |
| in vivo | In xenografts of both breast and colorectal cancers, ICEC0942 has substantial anti-tumor effects. For pharmacokinetics, CD1 male mice are treated intravenously (IV), subcutaneously (SC) or by oral gavage (PO) with 10 mg/kg ICEC0942. In plasma, ICEC0942 levels decline in a bi-phasic manner, indicating rapid distribution into tissues. Following IV administration of ICEC0942 at 10 mg/kg in male CD1 mice Cl(plasma) is calculated at 78 ml.min/kg. Blood/plasma ratio (Bl/Pl) is 1.81. ICEC0942 has a half-life of 1.9 hrs, a moderate half-life in this species. Only a small proportion (13.5%) of ICEC0942 is metabolized after 2 and 4 hour following a single PO administration (100 mg/kg). Comparing exposure (AUCt) after single PO and IV administration at 10 mg/kg, oral bioavailability (F%) is calculated at 30%. Median Tmax for PO administration is 2 hours and is unaffected by increasing dose. Over this dose range, Cmax is linearly associated with dose, as is the total exposure over time (AUCt). In tumor-bearing mice, there is appreciable accumulation of ICEC0942 in tumors 6-hours post administration. ICEC0942 levels in tumors lag behind plasma levels[1]. |
プロトコル(参考用のみ)
| 細胞アッセイ | 細胞株 | HCT116 cells |
|---|---|---|
| 濃度 | 0.1, 1, 10 μM | |
| 反応時間 | 4, 8, 16, 24 h | |
| 実験の流れ | -- |
|
| 動物実験 | 動物モデル | seven-week old female nu/nu-BALB/c athymic nude mice with tumour xenograft |
| 投薬量 | 100 mg/kg/day | |
| 投与方法 | PO |
Selleckの高級品が、幾つかの出版された研究調査結果(以下を含む)で使われた:
| Genome-wide mapping of cancer dependency genes and genetic modifiers of chemotherapy in high-risk hepatoblastoma [ Nat Commun, 2023, 14(1):4003] | PubMed: 37414763 |
| Recurrent Mutations in Cyclin D3 Confer Clinical Resistance to FLT3 Inhibitors in Acute Myeloid Leukemia [ Clin Cancer Res, 2021, 10.1158/1078-0432.CCR-20-3458] | PubMed: 34103301 |
長期の保管のために-20°Cの下で製品を保ってください。
人間や獣医の診断であるか治療的な使用のためにでない。
各々の製品のための特定の保管と取扱い情報は、製品データシートの上で示されます。大部分のSelleck製品は、推薦された状況の下で安定です。製品は、推薦された保管温度と異なる温度で、時々出荷されます。長期の保管のために必要とされてそれと異なる温度で、多くの製品は、短期もので安定です。品質を維持するが、夜通しの積荷のために最も経済的な貯蔵状況を用いてあなたの送料を保存する状況の下に、製品が出荷されることを、我々は確実とします。製品の受領と同時に、製品データシートの上で貯蔵推薦に従ってください。
