|
受注:045-509-1970 |
技術サポート:tech@selleck.co.jp 平日9:00〜18:00 1営業日以内にご連絡を差し上げます |
化学情報
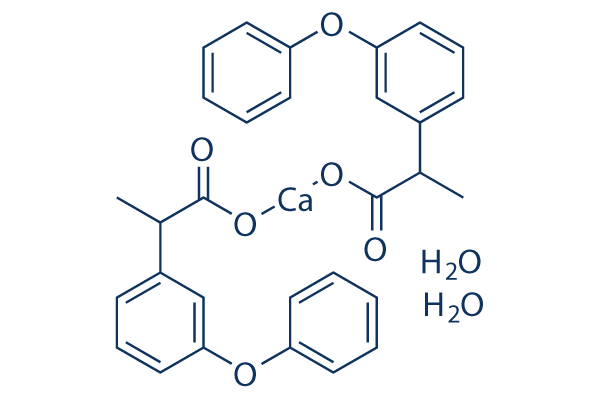
|
Synonyms | Fenoprofen calcium salt dihydrate,Feprona dihydrate,Progesic dihydrate | Storage (From the date of receipt) |
3 years -20°C powder 1 years -80°C in solvent |
|||||||
| 化学式 | C30H30CaO8 |
||||||||||
| 分子量 | 558.63 | CAS No. | 71720-56-4, 53746-45-5 | ||||||||
| Solubility (25°C)* | 体外 | DMSO | 48 mg/mL (85.92 mM) | ||||||||
| Ethanol | 18 mg/mL (32.22 mM) | ||||||||||
| Water | Insoluble | ||||||||||
| 体内 (毎回新しく調製した物を用意してください) |
|
||||||||||
|
* <1 mg/ml means slightly soluble or insoluble. * Please note that Selleck tests the solubility of all compounds in-house, and the actual solubility may differ slightly from published values. This is normal and is due to slight batch-to-batch variations. |
|||||||||||
溶剤液(一定の濃度)を調合する
生物活性
| 製品説明 | Fenoprofen calcium hydrate(Fenoprofen calcium salt dihydrate,Feprona dihydrate,Progesic dihydrate) は非ステロイド性抗炎症薬(NSAID)です。 |
|---|---|
| in vitro | Fenoprofen is a more potent inhibitor of collagen-induced platelet aggregation than either aspirin or phenylbutazone. Fenoprofen inhibits the formation of palmitoyl-CoA in both microsomal and peroxisomal fractions, and inhibits the beta-oxidation of lignoceric acid and cerotic acid in rat hepatocytes. Fenoprofen exhibits modest antiproliferative activity against HT-29, DID-1, and SW480 cells with IC50 of 240 μM, 300 μM, and 360 μM, respectively. Fenoprofen (0.1 mM) is an efficient activator of peroxisome proliferator-activated receptor gamma (PPARγ), activating the receptor to a degree comparable to that obtained with the PPARγ ligands BRL49653 and 15-deoxy-D12,14-PGJ2 and the peroxisome proliferator Wy14643. Fenoprofen is also an efficacious activator of PPARα, activating the receptor to a degree comparable to that obtained with the strong peroxisome proliferator Wy14643. Consistently, Fenoprofen treatment promotes lipogenesis in C3H10T1/2 cells. Although Fenoprofen displays only modest antiproliferative activity, Fenoprofen amides can potently induce cell cycle arrest at the G1 phase, as well as apoptosis, probably because of a greater lipophilicity and/or better cell uptake. |
| in vivo | Oral administration of Fenoprofen at 50 mg/kg potently inhibits thrombus formation by 47%, whereas a dose of 200 mg/kg of aspirin is required to reduce thrombus formation 21 %. Similar to indomethacin, adminstration of Fenoprofen inhibits prostaglandin synthesis. In rats with type II collagen-induced arthritis, Fenoprofen treatment at 40 mg/kg/day partially suppresses the paw swelling, but has no significant effect on humoral and cellular responses. Administration of Fenoprofen depresses the rebound contraction, thus transforming the brisk relaxant response, elicited by vagal stimulation or ATP, into long-lasting relaxation. Administration of Fenoprofen causes a strong and dose-related induction of peroxisomal palmitoyl-CoA oxidase, and of carnitine acyltransferase and acyl-CoA hydrolase activities in liver homogenates of mice fed diets. Hepatic catalase activity is significantly increased in mice fed the diet with 0.05 and 0.1% fenoprofen but not the 1% fenoprofen-containing diet. |
プロトコル(参考用のみ)
| 細胞アッセイ | 細胞株 | HT-29, DID-1, and SW480 |
|---|---|---|
| 濃度 | Dissolved in DMSO, final concentrations ~1 mM | |
| 反応時間 | 6 days | |
| 実験の流れ | Cells are exposed to various concentrations of Fenoprofen for 6 days. Cell number is determined using the SRB colonimetnic protein stain assay. After 6 days of culture, cells are fixed by the addition of cold trichloroacetic acid to a final concentration of 10%. Plates are incubated at 4 °C for 1 hour, then the supernatant is aspirated and the plates are washed with deionized water. SRB solution is formulated to 0.4% w/v in 1% acetic acid; 100 μL is added to each well and the plates are incubated for 10 minutes at room temperature. Unbound SRB is removed by washing with 1% acetic acid followed by air drying. Bound stain is solubilized with 50 mM unbuffered Tris and absorbance is read by an automated spectrophotometer at a single wavelength of 540 nm. |
|
| 動物実験 | 動物モデル | Guinea pigs with induced thrombus formation |
| 投薬量 | ~200 mg/kg | |
| 投与方法 | Orally |
参考
|
Selleckの高級品が、幾つかの出版された研究調査結果(以下を含む)で使われた:
| Salivary gland tissue chip screening identifies candidate radioprotective drugs [ Communications Medicine, 2025, 420] | PubMed: 41068535 |
| Identification and drug-induced reversion of molecular signatures of Alzheimers disease onset and progression in AppNL-G-F, AppNL-F, and 3xTg-AD mouse models [ Genome Medicine, 2021, 168] | PubMed: nan |
| Identification and drug-induced reversion of molecular signatures of Alzheimer's disease onset and progression in AppNL-G-F, AppNL-F, and 3xTg-AD mouse models [ Genome Med, 2021, 13(1):168] | PubMed: 34702310 |
長期の保管のために-20°Cの下で製品を保ってください。
人間や獣医の診断であるか治療的な使用のためにでない。
各々の製品のための特定の保管と取扱い情報は、製品データシートの上で示されます。大部分のSelleck製品は、推薦された状況の下で安定です。製品は、推薦された保管温度と異なる温度で、時々出荷されます。長期の保管のために必要とされてそれと異なる温度で、多くの製品は、短期もので安定です。品質を維持するが、夜通しの積荷のために最も経済的な貯蔵状況を用いてあなたの送料を保存する状況の下に、製品が出荷されることを、我々は確実とします。製品の受領と同時に、製品データシートの上で貯蔵推薦に従ってください。
