- 阻害剤
- 研究分野別
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- 化合物ライブラリー
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-II
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleck.co.jp to customize your library.
You could select:
- 抗体
- 新製品
- お問い合わせ
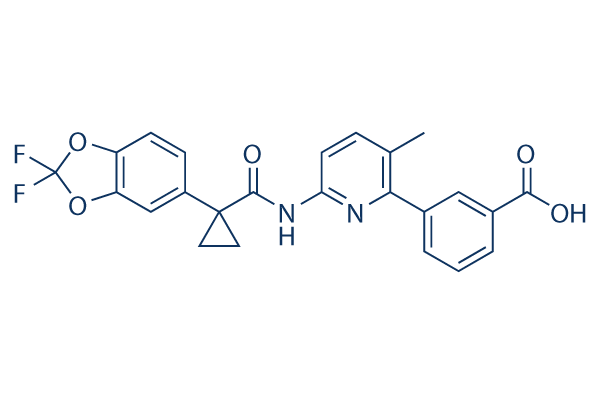
Lumacaftor (VX-809)
別名:VRT 826809
Lumacaftor (VX-809, VRT 826809) acts to correct CFTR mutations common in cystic fibrosis by increasing mutant CFTR (F508del-CFTR) maturation,EC50 of 0.1 μM in fisher rat thyroid cells. Phase 3.
CAS No. 936727-05-8
文献中Selleckの製品使用例(232)
製品安全説明書
現在のバッチを見る:
純度:
99.96%
99.96
Lumacaftor (VX-809)関連製品
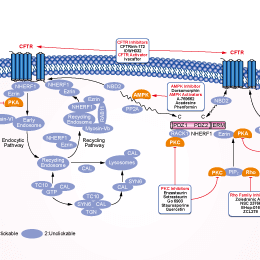
シグナル伝達経路
CFTR阻害剤の選択性比較
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | 活性情報 | PMID |
|---|---|---|---|---|---|
| CFBE41o | Corrector assay | 1 uM | 24 hrs | Corrector activity at CFTR F508-del mutant (unknown origin) expressed in human CFBE41o cells harboring HS-YFP assessed as increase in matured protein levels at cell surface at 1 uM after 24 hrs by electrophoretic mobility assay | 29272749 |
| CFBE41o | Corrector assay | 1 uM | 24 hrs | Corrector activity at CFTR F508del mutant (unknown origin) expressed in human CFBE41o cells assessed as increase in size of cAMP-dependent current at 1 uM after 24 hrs measured at +100 mV by whole cell patch clamp assay | 26041577 |
| CFBE41o | Corrector assay | 24 hrs | Corrector activity at CFTR F508-del mutant (unknown origin) expressed in human CFBE41o cells harboring HS-YFP preincubated for 24 hrs followed by forskolin/genistein stimulation for 30 mins by fluorescence assay, EC50 = 2.5704 μM. | 29272749 | |
| FRT | Corrector assay | 25 mins | Corrector activity at human CFTR F508 deletion mutant expressed in FRT cells incubated for 25 mins with forskolin by YFP-based fluorescence analysis relative to control, EC50 = 2.6 μM. | 26561003 | |
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for TC32 cells | 29435139 | ||
| U-2 OS | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for U-2 OS cells | 29435139 | ||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | ||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | ||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | ||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | ||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | ||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | ||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 29435139 | ||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | ||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | ||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for BT-12 cells | 29435139 | ||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | ||
| fibroblast cells | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for control Hh wild type fibroblast cells | 29435139 | ||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | ||
| Rh30 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh30 cells | 29435139 | ||
| U-2 OS | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for U-2 OS cells | 29435139 | ||
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for Rh41 cells | 29435139 | ||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Confirmatory screen for RD cells | 29435139 | ||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | ||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | ||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | ||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | ||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | ||
| HBE | Corrector assay | Corrector activity at CFTR F508del/F508del mutant in primary HBE cells assessed as increase in chloride ion current across apical membrane measured 18 to 24 hrs post compound treatment on basolateral side of cells in presence of channel potentiator GLPG18 | 29251932 | ||
| CFBE41o | Corrector assay | Corrector activity at CFTR F508del mutant (unknown origin) expressed in human CFBE41o cells assessed as increase in fully glycosylated protein by western blot analysis | 26041577 | ||
| 他の多くの細胞株試験データをご覧になる場合はこちらをクリックして下さい | |||||
生物活性
| 製品説明 | Lumacaftor (VX-809, VRT 826809) acts to correct CFTR mutations common in cystic fibrosis by increasing mutant CFTR (F508del-CFTR) maturation,EC50 of 0.1 μM in fisher rat thyroid cells. Phase 3. | ||
|---|---|---|---|
| 特性 | Higher specificity and efficacy relative to other CFTR defect drugs. | ||
| Targets |
|
| In Vitro | ||||
| In vitro |
Lumacaftor (VX-809) acts at the level of the ER to allow a fraction of the F508del-CFTR to adopt a properly folded form, to exit the ER and mobilize to the cell surface for normal functioning. In Fischer rat thyroid (FRT) cells expressing F508del-CFTR, this compound treatment significantly improves F508del-CFTR maturation by 7.1 fold with an EC50 of 0.1 μM, and enhances F508del-CFTR-mediated chloride transport by approximately 5 fold with EC50 of 0.5 μM, while VRT-768 has higher EC50 values of 7.9 μM and 16 μM, respectively. In HEK-293 cells expressing F508del-CFTR, it (3 μM) increases F508del-CFTR exit from the ER by 6 fold, reaching levels comparable to 34% of CFTR. In primary human bronchial epithelial (HBE) cells with F508del-CFTR mutation, the agent increases CFTR maturation and enhances chloride secretion with EC50 of 350 nM and 81 nM, respectively, more efficacious than Corr-4a and VRT-325. F508del-CFTR corrected by it exhibits single-channel open probability of 0.39 similar to normal CFTR of 0.40. Unlike VX-770, it is not a CFTR potentiator, as acute addition has no effect on F508del-CFTR function. In contrast to VRT-325 and Corr-4a, it does not improve the processing of the normal or mutant forms of hERG or P-gp, as well as other disease-causing mislocalized proteins, including α1-antitrypsin Z mutant (E342K-α1-AT) or N370S-β-glucosidase, suggesting that it is specific for CFTR. It in combination with VRT-325 or Corr-4a has additive effect on CFTR-mediated chloride transport in cultured F508del-HBE. |
|||
|---|---|---|---|---|
| Kinase Assay | F508del-CFTR maturation | |||
| FRT cells stably expressing F508del-CFTR are treated with increasing concentrations of Lumacaftor (VX-809) for 48 hours. After incubation, cells are harvested in ice-cold D-PBS solution (without calcium and magnesium) and pelleted at 1,000 × g at 4 °C. Cell pellets are lysed in 1% Nonidet P-40, 0.5% sodium deoxycholate, 200 mM NaCl, 10 mM Tris, pH 7.8, and 1 mM EDTA plus protease inhibitor mixture (1:250) for 30 minutes on ice. Lysates are spun for 10 minutes at 10,000 × g at 4 °C to pellet nuclei and insoluble material. Approximately 12 μg total protein is heated in Laemmli buffer with 5% β-mercaptoethanol at 37 °C for 5 minutes and loaded onto a 3% to 8% Tris-acetate gel. The gel is transferred to nitrocellulose and processed for Western blotting by using monoclonal CFTR antibody or polyclonal to GAPDH. Blots are developed by enhanced chemiluminescence. Quantification of the relative amounts of bands C and GAPDH is performed by using NIH ImageJ analysis of scanned films. | ||||
| 細胞実験 | 細胞株 | FRT (CFTR or F508del-CFTR), HEK-293 (CFTT or F508del-CFTR) , and HBE cells | ||
| 濃度 | Dissolved in DMSO, final concentrations ~0.1 mM | |||
| 反応時間 | 24 or 48 hours | |||
| 実験の流れ | Cells are exposed to various concentrations of Lumacaftor (VX-809) for 24 or 48 hours. Ussing chamber techniques are used to record the transepithelial current (IT) resulting from CFTR-mediated chloride transport. The single-channel activity of CFTR is measured by using excised inside-out membrane patch recordings. Immunoblot techniques using the monoclonal CFTR antibody are used to measure CFTR maturation in FRT, HEK-293, or HBE cells expressing CFTR or F508del-CFTR. |
|||
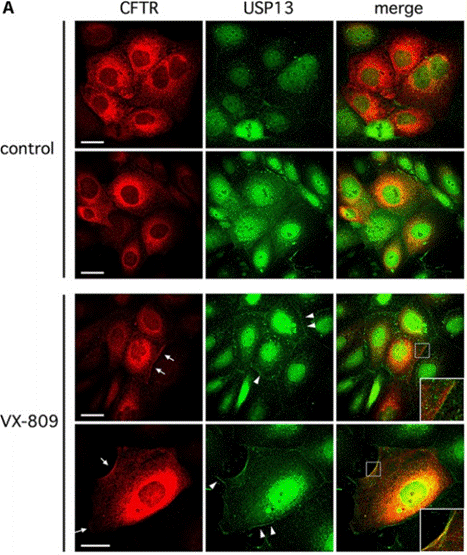
| 実験結果図 | Methods | Biomarkers | 結果図 | PMID |
| Immunofluorescence | CFTR / USP13 Cell surface kAE1 / kAE1 |
|
30618756 | |
| In Vivo | ||
| In Vivo |
Lumacaftor (VX-809; VRT 826809) is a CFTR modulator that corrects the folding and trafficking of CFTR protein. |
|
|---|---|---|
| 動物実験 | 動物モデル | Male Sprague– Dawley rats |
| 投与量 | 1 mg/kg | |
| 投与経路 | p.o. | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT03512119 | Completed | Cystic Fibrosis Homozygous for Phe 508 Del CFTR|Glucose Intolerance or Newly Diagnosis Diabetes |
University Hospital Strasbourg France |
February 11 2016 | -- |
| NCT02589236 | Completed | Cystic Fibrosis |
Nivalis Therapeutics Inc.|Medidata Solutions |
November 2015 | Phase 2 |
| NCT02514473 | Completed | Cystic Fibrosis |
Vertex Pharmaceuticals Incorporated |
July 2015 | Phase 3 |
| NCT01899105 | Completed | Cystic Fibrosis |
Vertex Pharmaceuticals Incorporated |
July 2013 | Phase 1 |
|
化学情報
| 分子量 | 452.41 | 化学式 | C24H18F2N2O5 |
| CAS No. | 936727-05-8 | SDF | Download Lumacaftor (VX-809) SDFをダウンロードする |
| Smiles | CC1=C(N=C(C=C1)NC(=O)C2(CC2)C3=CC4=C(C=C3)OC(O4)(F)F)C5=CC(=CC=C5)C(=O)O | ||
| 保管 | |||
|
In vitro |
DMSO : 90 mg/mL ( (198.93 mM); 吸湿したDMSOは溶解度を減少させます。新しいDMSOをご使用ください。) Ethanol : 6 mg/mL Water : Insoluble |
モル濃度計算器 |
|
in vivo Add solvents to the product individually and in order. |
投与溶液組成計算機 | |||||
実験計算
投与溶液組成計算機(クリア溶液)
ステップ1:実験データを入力してください。(実験操作によるロスを考慮し、動物数を1匹分多くして計算・調製することを推奨します)
mg/kg
g
μL
匹
ステップ2:投与溶媒の組成を入力してください。(ロット毎に適した溶解組成が異なる場合があります。詳細については弊社までお問い合わせください)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
計算結果:
投与溶媒濃度: mg/ml;
DMSOストック溶液調製方法: mg 試薬を μL DMSOに溶解する(濃度 mg/mL, 注:濃度が当該ロットのDMSO溶解度を超える場合はご連絡ください。 )
投与溶媒調製方法:Take μL DMSOストック溶液に μL PEG300,を加え、完全溶解後μL Tween 80,を加えて完全溶解させた後 μL ddH2O,を加え完全に溶解させます。
投与溶媒調製方法:μL DMSOストック溶液に μL Corn oil,を加え、完全溶解。
注意:1.ストック溶液に沈殿、混濁などがないことをご確認ください;
2.順番通りに溶剤を加えてください。次のステップに進む前に溶液に沈殿、混濁などがないことを確認してから加えてください。ボルテックス、ソニケーション、水浴加熱など物理的な方法で溶解を早めることは可能です。
技術サポート
ストックの作り方、阻害剤の保管方法、細胞実験や動物実験の際に注意すべき点など、製品を取扱う時に問い合わせが多かった質問に対しては取扱説明書でお答えしています。
他に質問がある場合は、お気軽にお問い合わせください。
* 必須