
- 阻害剤
- 研究分野別
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- 化合物ライブラリー
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- Customize Library(compound antibody kit)
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-II
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleck.co.jp to customize your library.
You could select:
- 抗体
- 新製品
- お問い合わせ
Integrase
亜型選択性的な製品
Integrase製品
- All (14)
- Integrase阻害剤 (14)
- 新製品
| 製品コード | 製品名称 | 製品説明 | 文献中Selleckの製品使用例 | お客様のフィードバック |
|---|---|---|---|---|
| S2005 | Raltegravir | Raltegravirは、WTおよびS217Q PFV Integraseに対する強力なIntegrase (IN)阻害剤であり、セル-フリーアッセイではそれぞれ90 nMおよび40 nMのIC50を示します。HCVポリメラーゼ、HIV逆転写酵素、HIV RNaseH、ヒトα-、β-、γ-ポリメラーゼなど、いくつかの関連するMg2+-依存性酵素に対するHIV-1 Integraseの選択性は1000倍を超えます。 |

|
|
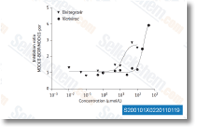
| S2001 | Elvitegravir | Elvitegravirは、HIV-1 IIIB、HIV-2 EHO、およびHIV-2 RODに対するHIV Integrase阻害剤であり、セルフリーアッセイでのIC50はそれぞれ0.7 nM、2.8 nM、1.4 nMです。 |
|
|
| S2667 | GSK1349572 (Dolutegravir) | Dolutegravirは、cell-freeアッセイにおいてIC50が2.7 nMである2つの金属結合型HIV integrase阻害剤であり、raltegravir耐性の特徴的な変異体であるY143R、Q148K、N155H、G140S/Q148Hに対しては中程度の活性を示します。 |
|
|
| S7766 | GSK1265744 (Cabotegravir) | Cabotegravir (GSK744, GSK1265744, S/GSK1265744) は、広範囲のHIVサブタイプに有効な長時間作用型HIVインテグラーゼ阻害剤であり、IC50 3.0 nMでHIV-1インテグラーゼ触媒鎖転移反応を阻害します。フェーズ2。 | ||
| S5245 | Raltegravir potassium | Raltegravir Potassium(MK-0518 potassium)は、経口生物学的利用能を有するRaltegravirのカリウム塩であり、初めて承認されたヒト免疫不全ウイルス1型(HIV-1)インテグラーゼ阻害剤です。 | ||
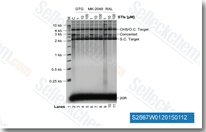
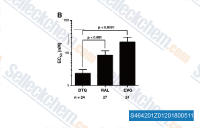
| S4642 | Dolutegravir Sodium | Dolutegravir Sodium (GSK-1349572A)は、IC50が2.7 nMのHIV integrase阻害剤です。 |
|
|
| S5911 | Bictegravir | Bictegravir(GS-9883)は、新規の強力な1日1回投与のブーストなしのHIV-1インテグラーゼ阻害剤です。 | ||
| S2765 | MK-2048 | MK-2048は、Integrase (IN)およびINR263Kの強力な阻害剤であり、それぞれ2.6 nMおよび1.5 nMのIC50を有します。 | ||
| E0161 | Bictegravir Sodium | Bictegravir (GS-9883) Sodium は、HIV-1 integrase の新規かつ強力な阻害剤であり、IN 鎖転移活性を特異的に標的とし、IC50 は 7.5 nM です。 |
||
| S1366 | BMS-707035 | BMS-707035は、IC50が15 nMのHIV-I integrase (IN)特異的阻害剤です。現在、第2相臨床試験中です。 | ||
| E4468 | Cabotegravir Sodium | Cabotegravir (GSK-1265744) Sodiumは、経口活性のある長時間作用型のHIVインテグラーゼ鎖転移阻害剤である。また、有機アニオントランスポーター1/3 (OAT1/OAT3)を阻害し、OAT3およびOAT1に対するIC50値はそれぞれ0.41 μMおよび0.81 μMである。 | ||
| S4187 | Salicylanilide | Salicylanilide (WR10019、2-ヒドロキシベンズアニリド) は、抗ウイルス作用、抗菌作用(抗マイコバクテリア作用を含む)、抗真菌作用など、幅広い生物学的活性を持つ化合物のグループです。 | ||
| E0459 | Robinetin | Robinetinは、用量依存的にHIVインテグラーゼの切断と統合を阻害し、Proteus vulgarisのDNA合成を、S. aureusのRNA合成を阻害し、さらに卵黄ホスファチジルコリン(EYPC)膜脂質過酸化とヘモグロビンA(HbA)の糖化を高効率で阻害します。 | ||
| S0235 | Lavendustin B | Lavendustin Bは、HIV-1インテグラーゼ (IN)とその同族の細胞補因子である水晶体上皮由来増殖因子 (LEDGF/p75)との相互作用を阻害します。Lavendustin Bはチロシンキナーゼ阻害剤であり、グルコーストランスポーター1 (Glut1)の競合的阻害剤でもあります。 | ||
| S2005 | Raltegravir | Raltegravirは、WTおよびS217Q PFV Integraseに対する強力なIntegrase (IN)阻害剤であり、セル-フリーアッセイではそれぞれ90 nMおよび40 nMのIC50を示します。HCVポリメラーゼ、HIV逆転写酵素、HIV RNaseH、ヒトα-、β-、γ-ポリメラーゼなど、いくつかの関連するMg2+-依存性酵素に対するHIV-1 Integraseの選択性は1000倍を超えます。 |

|
|
| S2001 | Elvitegravir | Elvitegravirは、HIV-1 IIIB、HIV-2 EHO、およびHIV-2 RODに対するHIV Integrase阻害剤であり、セルフリーアッセイでのIC50はそれぞれ0.7 nM、2.8 nM、1.4 nMです。 |

|
|
| S2667 | GSK1349572 (Dolutegravir) | Dolutegravirは、cell-freeアッセイにおいてIC50が2.7 nMである2つの金属結合型HIV integrase阻害剤であり、raltegravir耐性の特徴的な変異体であるY143R、Q148K、N155H、G140S/Q148Hに対しては中程度の活性を示します。 |

|
|
| S7766 | GSK1265744 (Cabotegravir) | Cabotegravir (GSK744, GSK1265744, S/GSK1265744) は、広範囲のHIVサブタイプに有効な長時間作用型HIVインテグラーゼ阻害剤であり、IC50 3.0 nMでHIV-1インテグラーゼ触媒鎖転移反応を阻害します。フェーズ2。 | ||
| S5245 | Raltegravir potassium | Raltegravir Potassium(MK-0518 potassium)は、経口生物学的利用能を有するRaltegravirのカリウム塩であり、初めて承認されたヒト免疫不全ウイルス1型(HIV-1)インテグラーゼ阻害剤です。 | ||
| S4642 | Dolutegravir Sodium | Dolutegravir Sodium (GSK-1349572A)は、IC50が2.7 nMのHIV integrase阻害剤です。 |

|
|
| S5911 | Bictegravir | Bictegravir(GS-9883)は、新規の強力な1日1回投与のブーストなしのHIV-1インテグラーゼ阻害剤です。 | ||
| S2765 | MK-2048 | MK-2048は、Integrase (IN)およびINR263Kの強力な阻害剤であり、それぞれ2.6 nMおよび1.5 nMのIC50を有します。 | ||
| E0161 | Bictegravir Sodium | Bictegravir (GS-9883) Sodium は、HIV-1 integrase の新規かつ強力な阻害剤であり、IN 鎖転移活性を特異的に標的とし、IC50 は 7.5 nM です。 |
||
| S1366 | BMS-707035 | BMS-707035は、IC50が15 nMのHIV-I integrase (IN)特異的阻害剤です。現在、第2相臨床試験中です。 | ||
| E4468 | Cabotegravir Sodium | Cabotegravir (GSK-1265744) Sodiumは、経口活性のある長時間作用型のHIVインテグラーゼ鎖転移阻害剤である。また、有機アニオントランスポーター1/3 (OAT1/OAT3)を阻害し、OAT3およびOAT1に対するIC50値はそれぞれ0.41 μMおよび0.81 μMである。 | ||
| S4187 | Salicylanilide | Salicylanilide (WR10019、2-ヒドロキシベンズアニリド) は、抗ウイルス作用、抗菌作用(抗マイコバクテリア作用を含む)、抗真菌作用など、幅広い生物学的活性を持つ化合物のグループです。 | ||
| E0459 | Robinetin | Robinetinは、用量依存的にHIVインテグラーゼの切断と統合を阻害し、Proteus vulgarisのDNA合成を、S. aureusのRNA合成を阻害し、さらに卵黄ホスファチジルコリン(EYPC)膜脂質過酸化とヘモグロビンA(HbA)の糖化を高効率で阻害します。 | ||
| S0235 | Lavendustin B | Lavendustin Bは、HIV-1インテグラーゼ (IN)とその同族の細胞補因子である水晶体上皮由来増殖因子 (LEDGF/p75)との相互作用を阻害します。Lavendustin Bはチロシンキナーゼ阻害剤であり、グルコーストランスポーター1 (Glut1)の競合的阻害剤でもあります。 |
