- 阻害剤
- 研究分野別
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- 化合物ライブラリー
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- Customize Library(compound antibody kit)
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-II
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleck.co.jp to customize your library.
You could select:
- 抗体
- 新製品
- お問い合わせ
KW-2449
KW-2449は、IC50が6.6 nMで主にFLT3を標的とする多標的阻害剤であり、FGFR1、Bcr-Abl、Aurora Aに対して中程度の効力があります。PDGFRβ、IGF-1R、EGFRにはほとんど影響がありません。第1相。
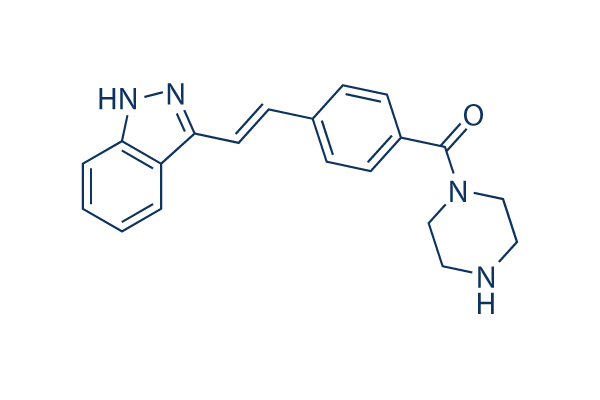
CAS No. 1000669-72-6
文献中Selleckの製品使用例(12)
カスタマーフィードバック1例
製品安全説明書
現在のバッチを見る:
S215802
DMSO]
67 mg/mL]
false]
Ethanol]
67 mg/mL]
false]
Water]
Insoluble]
false
純度:
99.77%
99.77
KW-2449関連製品
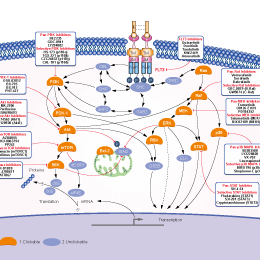
シグナル伝達経路
FLT3阻害剤の選択性比較
生物活性
| 製品説明 | KW-2449は、IC50が6.6 nMで主にFLT3を標的とする多標的阻害剤であり、FGFR1、Bcr-Abl、Aurora Aに対して中程度の効力があります。PDGFRβ、IGF-1R、EGFRにはほとんど影響がありません。第1相。 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 特性 | Investigated as a FLT3 inhibitor in clinical trials, with others in early development. | |||||||||||
| Targets |
|
| In Vitro | ||||
| In vitro | KW-2449, a multikinase inhibitor of FLT3, ABL, ABL-T315I, and Aurora kinase, is under investigation to treat leukemia patients. This compound shows the potent growth inhibitory effects on leukemia cells with FLT3 mutations by inhibition of the FLT3 kinase, resulting in the down-regulation of phosphorylated-FLT3/STAT5, G1 arrest, and apoptosis. Oral administration of this chemical shows dose-dependent and significant tumor growth inhibition in FLT3-mutated xenograft model with minimum bone marrow suppression. In FLT3 wild-type human leukemia, it induces the reduction of phosphorylated histone H3, G2/M arrest, and apoptosis. In Imatinib-resistant leukemia, this agent contributes to release of the resistance by the simultaneous down-regulation of BCR/ABL and Aurora kinases. The inhibitory activity of this compound is not affected by the presence of human plasma protein, such as α1-acid glycoprotein. It has potent growth inhibitory activity against various types of leukemia by several mechanisms of action. This inhibitor has significant activity and warrants clinical study in leukemia patients with FLT3 mutations as well as Imatinib-resistant mutations. Phosphorylation levels of FLT3 and STAT5 are decreased by this chemical in a dose-dependent manner. In addition, it potently inhibits ABL-T315I, which is associated with IM resistance, with IC50 of 4 nM. On the other hand, this compound has little effect on PDGFRβ, IGF-1R, EGFR, and various serine/threonine kinases even at a concentration of 1 μM. It has the potent growth inhibitory activities against not only FLT3/ITD-expressing leukemia cells but also FLT3/KDM-activated and wild-type FLT3-overexpressing leukemia cells. In accordance with growth inhibitory effect, this agent suppresses the phosphorylations of FLT3 (P-FLT3) and its downstream molecule phospho-STAT5 (P-STAT5) in MOLM-13 cells in a dose-dependent manner. Furthermore, it increases the percentage of cells in the G1 phase of the cell cycle and reciprocally reduces the percentage of cells in the S phase, resulting in the increase of apoptotic cell population. This compound can dephosphorylate constitutively active WT-FLT3 kinase but not inhibit the proliferation of leukemia cells if they are not mainly addicted to FLT3 the kinase. It is rapidly absorbed and converted to a major metabolite M1.Preclinical studies reveal that this chemical is converted by monoamine oxidase-B (MAO-B) and aldehyde oxidase into its major metabolite M1.This agent mediates cytotoxicity thru inhibition of FLT3/ITD.It is a direct inhibitor of FLT3 and induces inhibition of its downstream target STAT5. This compound interacts synergistically with HDACIs to induce apoptosis in Ph+ CML cells in a time- and concentration-dependent manner. It synergistically enhances the lethality of vorinostat/SNDX275 in CML cells.This inhibitor regimens are active against additional IM-resistant Bcr/Abl+ leukemia cells. It moderately reduces phosphorylation of histone H3, an indicator of Aurora B activity, in nocodozole-treated K562 cells. | |||
|---|---|---|---|---|
| Kinase Assay | FLT3 phosphorylation | |||
| Leukemia cells are washed in phosphate-buffered saline (PBS), then lysed by resuspending the cells in lysis buffer (20 mM Tris pH 7.4, 100 mM NaCl, 1% Igepal, 1 mM EDTA, 2 mM NaVO4, plus Complete protease inhibitor KW-2449 for 30 minutes while rocking. The extract is clarified by centrifugation at 1.6 × 104 g and the supernatant is assayed for protein (Bio-Rad). A 50-μg aliquot is removed as a whole-cell lysate for analysis of STAT5, and the remainder is used for immunoprecipitation with anti-FLT3. Anti-FLT3 antibody is added to the extract for overnight incubation, then protein A sepharose is added for 2 additional hours. Separate sodium dodecyl sulfate–polyacrylamide electrophoresis (SDS-PAGE) gels for whole-cell lysate and immunoprecipates are run in parallel. After transfer to Immobilon membranes, immunoblotting is performed with antiphosphotyrosine antibody (4G10) to detect phosphorylated FLT3 or, for the whole-cell lysate gels, with a rat monoclonal antibody against phosphorylated STAT5 (residue Y694) then stripped and reprobed with anti-FLT3 antibody to measure total FLT3. Proteins are visualized using chemiluminescence, exposed on Kodak BioMax XAR film, developed, and scanned using a Bio-Rad GS800 densitometer. The concentration of this compound for which the phosphorylation of FLT3 or STAT5 is inhibited to 50% of its baseline (IC50) is determined using linear regression analysis of the dose response curves. For direct analysis of FLT3 and STAT5 in circulating blasts, peripheral blood is collected in heparinized tubes and promptly chilled on ice. Samples are centrifuged for 10 minutes at 900 g, at 4 °C. The plasma is removed and stored frozen at −80 °C. The buffy coat is carefully transferred to ice-cold PBS, layered onto chilled Ficoll-Hypaque, and centrifuged for 5 minutes at 600 g, at 4 °C. All subsequent steps are carried out at 4 °C. Mononuclear cells are collected and washed rapidly once in red blood cell lysis buffer (0.155 M NH4Cl, 0.01 M KHCO3, 0.1 mM EDTA), then washed once in PBS. Cells are then lysed as described for FLT3 and STAT5 analysis. | ||||
| 細胞実験 | 細胞株 | MOLM-13 and RS4;11 cells | ||
| 濃度 | 33nM, 75nM, 0.1μM, 0.3μM and 0.15μM | |||
| 反応時間 | 24, 48, and 72 hours | |||
| 実験の流れ | Cell viability is determined by the sodium 3′-[1-(phenylaminocarbonyl)-3, 4-tetrazolium]-bis (4-methoxy-6-nitro) benzene sulfonic acid hydrate assay after incubation with or without KW-2449 for 72 hours at 37 °C. The number of viable cells is determined using the Cell Proliferation Kit II. For cell-cycle analysis, MOLM-13 and RS4;11 cells are treated with this compound. After 24, 48, and 72 hours of incubation at 37 °C, DNA contents are analyzed. Cell cycle distribution of K562, TCC-Y, and TCC/Ysr is analyzed 24 hours after treatment with this chemical or imatinib. | |||
| In Vivo | ||
| In Vivo | In the MOLM-13 tumor xenograft model, oral administration of KW-2449 for 14 days shows a potent and significant antitumor effect in a dose-dependent manner. | |
|---|---|---|
| 動物実験 | 動物モデル | CBySmn.CB17-Prkdsscid/J (BALB/C) mice are injected with BV173/E255K/Luc cl4 cells. |
| 投与量 | 32 mg/kg/day, 5 days/week | |
| 投与経路 | Orally (p.o.) administered | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT00346632 | Terminated | Acute Myelogenous Leukemia|Acute Lymphoblastic Leukemia|Myelodysplastic Syndromes|Chronic Myelogenous Leukemia |
Kyowa Kirin Inc. |
June 2006 | Phase 1 |
|
化学情報
| 分子量 | 332.4 | 化学式 | C20H20N4O |
| CAS No. | 1000669-72-6 | SDF | Download KW-2449 SDFをダウンロードする |
| Smiles | C1CN(CCN1)C(=O)C2=CC=C(C=C2)C=CC3=NNC4=CC=CC=C43 | ||
| 保管 | |||
|
In vitro |
DMSO : 67 mg/mL ( (201.56 mM); 吸湿したDMSOは溶解度を減少させます。新しいDMSOをご使用ください。) Ethanol : 67 mg/mL Water : Insoluble |
モル濃度計算器 |
|
in vivo Add solvents to the product individually and in order. |
投与溶液組成計算機 | |||||
実験計算
投与溶液組成計算機(クリア溶液)
ステップ1:実験データを入力してください。(実験操作によるロスを考慮し、動物数を1匹分多くして計算・調製することを推奨します)
mg/kg
g
μL
匹
ステップ2:投与溶媒の組成を入力してください。(ロット毎に適した溶解組成が異なる場合があります。詳細については弊社までお問い合わせください)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
計算結果:
投与溶媒濃度: mg/ml;
DMSOストック溶液調製方法: mg 試薬を μL DMSOに溶解する(濃度 mg/mL, 注:濃度が当該ロットのDMSO溶解度を超える場合はご連絡ください。 )
投与溶媒調製方法:Take μL DMSOストック溶液に μL PEG300,を加え、完全溶解後μL Tween 80,を加えて完全溶解させた後 μL ddH2O,を加え完全に溶解させます。
投与溶媒調製方法:μL DMSOストック溶液に μL Corn oil,を加え、完全溶解。
注意:1.ストック溶液に沈殿、混濁などがないことをご確認ください;
2.順番通りに溶剤を加えてください。次のステップに進む前に溶液に沈殿、混濁などがないことを確認してから加えてください。ボルテックス、ソニケーション、水浴加熱など物理的な方法で溶解を早めることは可能です。
技術サポート
ストックの作り方、阻害剤の保管方法、細胞実験や動物実験の際に注意すべき点など、製品を取扱う時に問い合わせが多かった質問に対しては取扱説明書でお答えしています。
他に質問がある場合は、お気軽にお問い合わせください。
* 必須