- 阻害剤
- 研究分野別
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- 化合物ライブラリー
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- Customize Library(compound antibody kit)
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-II
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleck.co.jp to customize your library.
You could select:
- 抗体
- 新製品
- お問い合わせ
Fenebrutinib (GDC-0853)
Fenebrutinib (GDC-0853)は、強力で選択的な非共有結合性ブルトン型チロシンキナーゼ(BTK)阻害剤であり、Btkに対するKi値は0.91 nMで、3つのオフターゲット(Bmx:153倍、Fgr:168倍、Src:131倍)に対して100倍以上の選択性を示します。
CAS No. 1434048-34-6
文献中Selleckの製品使用例(4)
製品安全説明書
現在のバッチを見る:
S842101
DMSO]
8 mg/mL]
false]
Water]
Insoluble]
false]
Ethanol]
Insoluble]
false
純度:
99.91%
99.91
Fenebrutinib (GDC-0853)関連製品
シグナル伝達経路
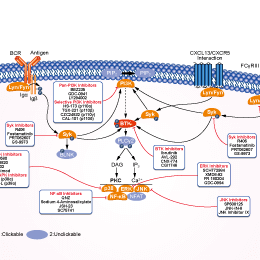
BTK阻害剤の選択性比較
生物活性
| 製品説明 | Fenebrutinib (GDC-0853)は、強力で選択的な非共有結合性ブルトン型チロシンキナーゼ(BTK)阻害剤であり、Btkに対するKi値は0.91 nMで、3つのオフターゲット(Bmx:153倍、Fgr:168倍、Src:131倍)に対して100倍以上の選択性を示します。 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Targets |
|
| In Vitro | ||||
| In vitro |
When tested at 1 μM against a broad panel of human kinase biochemical assays, Fenebrutinib (GDC-0853) inhibits only 3 of 286 off-target kinases. Based on the determined IC50 values, the selectivity for Btk is >100-fold against each of these 3 off-targets: Bmx (153-fold), Fgr (168-fold), and Src (131-fold). This compound blocks both B-cell BCR and monocyte FcγR signaling. In in vitro biochemical Btk enzyme assay, it displays an average residence time with Btk of 18.3 ± 2.8 hours. It blocks cellular autophosphorylation of WT Btk and the C481S mutant. CLL (chronic lymphocytic leukemia) cells treated with GDC-0853 in vitro before BCR stimulation demonstrate reduced levels of BTK phosphorylation and diminished activation of downstream targets including PLCγ2, AKT, and ERK. It inhibits NF-κB–dependent transcription, reduces activation, and impairs migration. This compound lacks inhibition of EGFR and ITK in cellular system and does not affect T-cell receptor activation. |
|||
|---|---|---|---|---|
| Kinase Assay | Kinase selectivity | |||
| Fenebrutinib (GDC-0853) kinase selectivity is assessed at a concentration of 1 µM in a panel of up to 287 recombinant human kinase activity and binding assays, including cytoplasmic and receptor tyrosine kinases, serine/threonine kinases, and lipid kinases. The kinase activity assays measure peptide phosphorylation or ADP production while the binding assays monitored displacement of ATP sitebinding probes. The ATP concentrations used in the activity assays are typically within 2-fold of the experimentally determined apparent Michaelis constant (Kmapp) value for each kinase while the competitive binding tracer concentrations used in the binding assays are generally within 3-fold of the experimentally determined dissociation constant (Kd) values. This compound is tested in duplicate against each kinase and the mean % Inhibition values are reported. For kinases that are inhibited by close to or greater than 80% at the test concentration, 10-point inhibitor titrations using the same assays are carried out in order to determine the inhibitor concentrations that caused 50% inhibition (IC50). | ||||
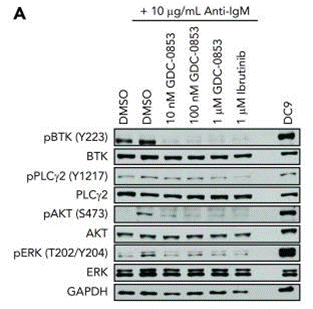
| 細胞実験 | 細胞株 | CLL cells | ||
| 濃度 | 1 μM | |||
| 反応時間 | 48 hours | |||
| 実験の流れ | Cells are treated with 1 µM Fenebrutinib (GDC-0853) for 48 hours and measured for viability by flow cytometry. | |||
| 実験結果図 | Methods | Biomarkers | 結果図 | PMID |
| Western blot | p-BTK / BTK / p-PLCγ2 / PLCγ2 / p-AKT / AKT / p-ERK / ERK |
|
30018078 | |
| In Vivo | ||
| In Vivo |
Fenebrutinib (GDC-0853) has moderate clearance of 27.4 mL/min/kg and excellent bioavailability (F=65%) in rats administered 0.2 mg/kg via intraperitoneal injection or 1 mg/kg PO. Its plasma clearance is 27.4 mL/min/kg, the volume of distribution (Vd) is 5.42 L/kg and the plasma half-life (t1/2) is 2.2 h. This compound also demonstrates favorable PK properties in dogs. The 3.8-hour half-life (Clp 10.9 mL/min/kg, Vd 2.96 L/kg) and high oral bioavailability (85%) also enable attainment of sufficient exposures in dog toxicology studies. It is well tolerated in both rats and dogs and displays an overall favorable safety profile. GDC-0853 is useful in treating rheumatoid arthritis and other B-cell or myeloid cell mediated autoimmune diseases. In a single ascending dose (SAD) study (0.5 mg to 600 mg) and multiple ascending dose (MAD) study for 14 days (250 mg BID to 500 mg QD), it is very well tolerated with no severe adverse events, no safety signals, and no dose limiting toxicities. It is well absorbed and had linear, doseproportional pharmacokinetics. In Sprague-Dawley (SD) rats, administration of this compound and other structurally diverse BTK inhibitors for 7 days or longer cause pancreatic lesions consisting of multifocal islet-centered hemorrhage, inflammation, fibrosis, and pigment-laden macrophages with adjacent lobular exocrine acinar cell atrophy, degeneration, and inflammation. Similar findings are not observed in mice or dogs at much higher exposures. |
|
|---|---|---|
| 動物実験 | 動物モデル | Sprague-Dawley, Wistar-Han and Fischer-344 rats (6 to 12 weeks old) |
| 投与量 | 5 or 10 mL/kg | |
| 投与経路 | p.o. | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT05119569 | Active not recruiting | Relapsing Multiple Sclerosis |
Hoffmann-La Roche |
March 1 2022 | Phase 2 |
| NCT04586023 | Active not recruiting | Relapsing Multiple Sclerosis |
Hoffmann-La Roche |
March 24 2021 | Phase 3 |
| NCT04586010 | Active not recruiting | Relapsing Multiple Sclerosis |
Hoffmann-La Roche |
March 17 2021 | Phase 3 |
| NCT03693625 | Terminated | Urticaria |
Genentech Inc. |
September 27 2018 | Phase 2 |
| NCT03596632 | Completed | Healthy Participants |
Hoffmann-La Roche |
July 27 2018 | Phase 1 |
|
化学情報
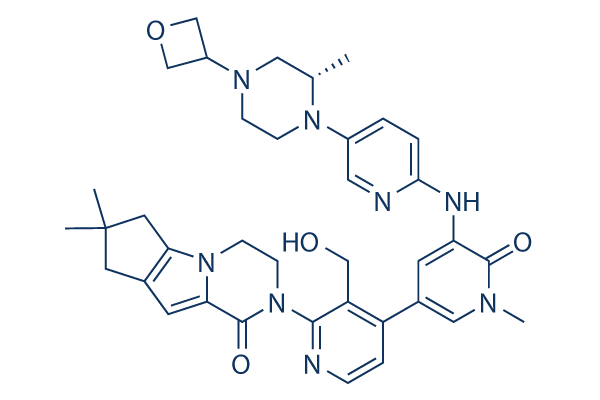
| 分子量 | 664.80 | 化学式 | C37H44N8O4 |
| CAS No. | 1434048-34-6 | SDF | -- |
| Smiles | CC1CN(CCN1C2=CN=C(C=C2)NC3=CC(=CN(C3=O)C)C4=C(C(=NC=C4)N5CCN6C7=C(CC(C7)(C)C)C=C6C5=O)CO)C8COC8 | ||
| 保管 | 3 years -20°C powder | ||
|
In vitro |
DMSO : 8 mg/mL ( (12.03 mM); 吸湿したDMSOは溶解度を減少させます。新しいDMSOをご使用ください。) Water : Insoluble Ethanol : Insoluble |
モル濃度計算器 |
|
in vivo Add solvents to the product individually and in order. |
投与溶液組成計算機 | |||||
実験計算
投与溶液組成計算機(クリア溶液)
ステップ1:実験データを入力してください。(実験操作によるロスを考慮し、動物数を1匹分多くして計算・調製することを推奨します)
mg/kg
g
μL
匹
ステップ2:投与溶媒の組成を入力してください。(ロット毎に適した溶解組成が異なる場合があります。詳細については弊社までお問い合わせください)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
計算結果:
投与溶媒濃度: mg/ml;
DMSOストック溶液調製方法: mg 試薬を μL DMSOに溶解する(濃度 mg/mL, 注:濃度が当該ロットのDMSO溶解度を超える場合はご連絡ください。 )
投与溶媒調製方法:Take μL DMSOストック溶液に μL PEG300,を加え、完全溶解後μL Tween 80,を加えて完全溶解させた後 μL ddH2O,を加え完全に溶解させます。
投与溶媒調製方法:μL DMSOストック溶液に μL Corn oil,を加え、完全溶解。
注意:1.ストック溶液に沈殿、混濁などがないことをご確認ください;
2.順番通りに溶剤を加えてください。次のステップに進む前に溶液に沈殿、混濁などがないことを確認してから加えてください。ボルテックス、ソニケーション、水浴加熱など物理的な方法で溶解を早めることは可能です。
技術サポート
ストックの作り方、阻害剤の保管方法、細胞実験や動物実験の際に注意すべき点など、製品を取扱う時に問い合わせが多かった質問に対しては取扱説明書でお答えしています。
他に質問がある場合は、お気軽にお問い合わせください。
* 必須