- 阻害剤
- 研究分野別
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- 化合物ライブラリー
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- Customize Library(compound antibody kit)
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-II
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleck.co.jp to customize your library.
You could select:
- 抗体
- 新製品
- お問い合わせ
AR-42
別名:HDAC-42
AR-42(HDAC-42)は、HDAC阻害剤であり、IC50は30 nMです。フェーズ1。
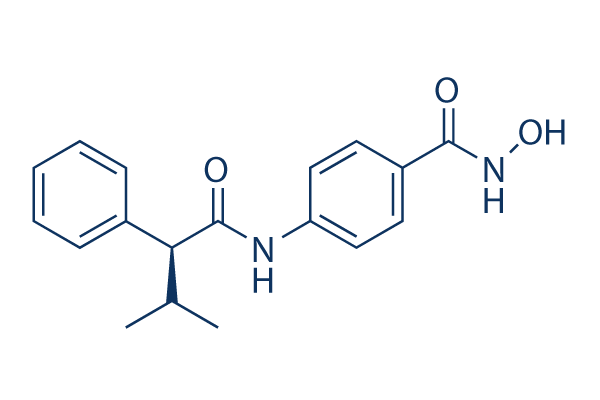
CAS No. 935881-37-1
文献中Selleckの製品使用例(30)
カスタマーフィードバック6例
製品安全説明書
現在のバッチを見る:
純度:
99.03%
99.03
AR-42関連製品
シグナル伝達経路
HDAC阻害剤の選択性比較
生物活性
| 製品説明 | AR-42(HDAC-42)は、HDAC阻害剤であり、IC50は30 nMです。フェーズ1。 | ||
|---|---|---|---|
| 特性 | Greater potency relative to SAHA. | ||
| Targets |
|
| In Vitro | ||||
| In vitro | AR-42 treatment induces histone hyperacetylation and p21WAF/CIP1 overexpression, and inhibits the growth of DU-145 cells with IC50 of 0.11 μM. This compound is potent in suppressing the proliferation of U87MG and PC-3 cells, in part, because of its ability to down-regulate Akt signaling. It inhibits the growth of PC-3 and LNCaP cells with IC50 of 0.48 μM and 0.3 μM, respectively. Compared to SAHA, this chemical exhibits distinctly superior apoptogenic potency, and causes markedly greater decreases in phospho-Akt, Bcl-xL, and survivin in PC-3 cells. This compound treatment induces growth inhibition, cell- cycle arrest, apoptosis, and activation of caspases-3/7 in malignant mast cell lines. It induces down-regulation of Kit via inhibition of Kit transcription, disassociation between Kit and heat shock protein 90 (HSP90), and up-regulation of HSP70. This treatment down-regulates the expression of p-Akt, total Akt, phosphorylated STAT3/5 (pSTAT3/5), and total STAT3/5. It potently inhibits the growth of JeKo-1, Raji, and 697 cells with IC50 of <0.61 μM. This compound also sensitizes CLL cells to TNF-Related Apoptosis Inducing Ligand (TRAIL), potentially through reduction of c-FLIP. This treatment also induces autophagy through downregulation of Akt/mTOR signaling and inducing ER stress in hepatocellular carcinoma (HCC) cells. | |||
|---|---|---|---|---|
| Kinase Assay | In vitro HDAC assay | |||
| HDAC activity is analyzed by using an HDAC assay kit. This assay is based on the ability of DU-145 nuclear extract, which is rich in HDAC activity, to mediate the deacetylation of the biotinylated [3H]-acetyl histone H4 peptide that is bound to streptavidin agarose beads. The release of [3H]-acetate into the supernatant is measured to calculate the HDAC activity. Sodium butyrate (0.25-1 mM) is used as a positive control. | ||||
| 細胞実験 | 細胞株 | DU-145 | ||
| 濃度 | Dissolved in DMSO, final concentrations ~2.5 μM | |||
| 反応時間 | 96 hours | |||
| 実験の流れ | Cells are exposed to varous concentrations of AR-42 for 96 hours. The medium is removed and replaced by 150 μL of 0.5 mg/mL of MTT in RPMI 1640 medium, and the cells are incubated in the CO2 incubator at 37 °C for 2 hours. Supernatants are removed from the wells, and the reduced MTT dye is solubilized with 200 μL/well of DMSO. Absorbance is determined on a plate reader at 570 nm. |
|||
| 実験結果図 | Methods | Biomarkers | 結果図 | PMID |
| Western blot | gp130 / p-STAT3 / STAT3 / p-AKT / AKT / p-MEK / MEK Cyclin D1 / p21 / p16 / Cyclin A / Cyclin B1 Act-H3 / Act-H3 / Act-tubulin p-Kit / Kit Notch1 / NICD / Nestin / Zeb-1 / BMI-1 |
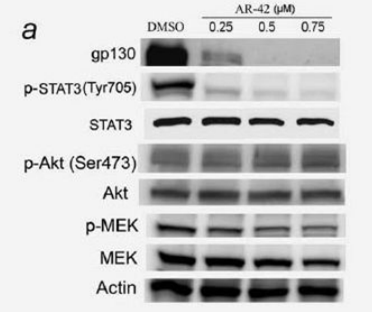
|
20824695 | |
| Growth inhibition assay | Cell viability |
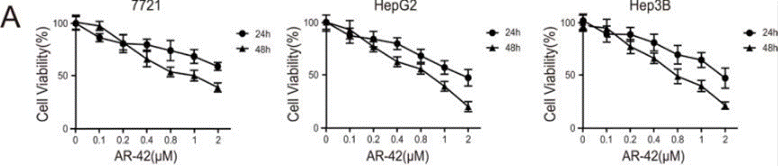
|
26993777 | |
| In Vivo | ||
| In Vivo | The growth of PC-3 tumor xenografts is suppressed by 52% and 67% after treatment with AR-42 at 25 mg/kg and 50 mg/kg, respectively, whereas SAHA at 50 mg/kg suppresses growth by 31%. In contrast to mice treated with SAHA, intratumoral levels of phospho-Akt and Bcl-xL are markedly reduced in this compound treated mice. In the transgenic adenocarcinoma of the mouse prostate (TRAMP) model, administration of this chemical not only decreases the severity of prostatic intraepithelial neoplasia (PIN) and completely prevents its progression to poorly differentiated carcinoma, but also shifts tumorigenesis to a more differentiated phenotype, suppressing absolute and relative urogenital tract weights by 86% and 85%, respectively. This agent significantly reduces leukocyte counts, and prolongs survival in three separate mouse models of B-cell malignancy without evidence of toxicity. | |
|---|---|---|
| 動物実験 | 動物モデル | Intact male NCr athymic nude mice inoculated s.c. with PC-3 cells |
| 投与量 | ~50 mg/kg/day | |
| 投与経路 | Orally | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02795819 | Terminated | Renal Cell Carcinoma|Soft Tissue Sarcoma|Metastatic Disease |
Virginia Commonwealth University|National Cancer Institute (NCI) |
July 8 2016 | Phase 1 |
| NCT02282917 | Terminated | Vestibular Schwannoma|Meningioma|Acoustic Neuroma|Neurofibromatosis Type 2 |
Massachusetts Eye and Ear Infirmary|Johns Hopkins University|Mayo Clinic|Stanford University|Ohio State University|Nationwide Children''s Hospital |
December 2015 | Early Phase 1 |
|
化学情報
| 分子量 | 312.36 | 化学式 | C18H20N2O3 |
| CAS No. | 935881-37-1 | SDF | Download AR-42 SDFをダウンロードする |
| Smiles | CC(C)C(C1=CC=CC=C1)C(=O)NC2=CC=C(C=C2)C(=O)NO | ||
| 保管 | |||
|
In vitro |
DMSO : 63 mg/mL ( (201.69 mM); 吸湿したDMSOは溶解度を減少させます。新しいDMSOをご使用ください。) Ethanol : 63 mg/mL Water : Insoluble |
モル濃度計算器 |
|
in vivo Add solvents to the product individually and in order. |
投与溶液組成計算機 | |||||
実験計算
投与溶液組成計算機(クリア溶液)
ステップ1:実験データを入力してください。(実験操作によるロスを考慮し、動物数を1匹分多くして計算・調製することを推奨します)
mg/kg
g
μL
匹
ステップ2:投与溶媒の組成を入力してください。(ロット毎に適した溶解組成が異なる場合があります。詳細については弊社までお問い合わせください)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
計算結果:
投与溶媒濃度: mg/ml;
DMSOストック溶液調製方法: mg 試薬を μL DMSOに溶解する(濃度 mg/mL, 注:濃度が当該ロットのDMSO溶解度を超える場合はご連絡ください。 )
投与溶媒調製方法:Take μL DMSOストック溶液に μL PEG300,を加え、完全溶解後μL Tween 80,を加えて完全溶解させた後 μL ddH2O,を加え完全に溶解させます。
投与溶媒調製方法:μL DMSOストック溶液に μL Corn oil,を加え、完全溶解。
注意:1.ストック溶液に沈殿、混濁などがないことをご確認ください;
2.順番通りに溶剤を加えてください。次のステップに進む前に溶液に沈殿、混濁などがないことを確認してから加えてください。ボルテックス、ソニケーション、水浴加熱など物理的な方法で溶解を早めることは可能です。
技術サポート
ストックの作り方、阻害剤の保管方法、細胞実験や動物実験の際に注意すべき点など、製品を取扱う時に問い合わせが多かった質問に対しては取扱説明書でお答えしています。
他に質問がある場合は、お気軽にお問い合わせください。
* 必須