- 阻害剤
- 研究分野別
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- 化合物ライブラリー
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- Customize Library(compound antibody kit)
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-II
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleck.co.jp to customize your library.
You could select:
- 抗体
- 新製品
- お問い合わせ
Ricolinostat (ACY-1215)
別名:Rocilinostat
Ricolinostat (ACY-1215, Rocilinostat) は、無細胞アッセイにおいて 5 nM の IC50 を持つ選択的 HDAC6 阻害剤です。HDAC1/2/3 (クラス I HDAC) よりも HDAC6 に対して 10 倍以上選択的で、HDAC8 に対してわずかな活性、HDAC4/5/7/9/11、Sirtuin1、Sirtuin2 に対しては最小限の活性しかありません。Ricolinostat (ACY-1215) は細胞増殖を抑制し、apoptosis を促進します。フェーズ 2。
CAS No. 1316214-52-4
文献中Selleckの製品使用例(71)
製品安全説明書
現在のバッチを見る:
純度:
99.96%
99.96
Ricolinostat (ACY-1215) と併用されることが多い化合物
It and JQ1 increase apoptosis, diminish the expression of c-MYC and BCL-2, and lower multiple myeloma cells proliferation.
Ricolinostat (ACY-1215) 関連製品
シグナル伝達経路
HDAC阻害剤の選択性比較
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | 活性情報 | PMID |
|---|---|---|---|---|---|
| SUP-B15 | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in imatinib-resistant human SUP-B15 cells assessed as increase in cleaved PARP expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 |
| SEM | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in human SEM cells assessed as increase in cleaved PARP expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 |
| HL60 | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in human HL60 cells assessed as increase in cleaved PARP expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 |
| SUP-B15 | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in imatinib-resistant human SUP-B15 cells assessed as increase in acetyl-histone H3 expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 |
| SEM | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in human SEM cells assessed as increase in acetyl-histone H3 expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 |
| HL60 | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in human HL60 cells assessed as increase in acetyl-histone H3 expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 |
| SUP-B15 | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in imatinib-resistant human SUP-B15 cells assessed as increase in acetyl-alpha tubulin expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 |
| SEM | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in human SEM cells assessed as increase in acetyl-alpha tubulin expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 |
| HL60 | Function assay | 0.1 to 10 uM | 24 hrs | Inhibition of HDAC6 in human HL60 cells assessed as increase in acetyl-alpha tubulin expression at 0.1 to 10 uM after 24 hrs by immunoblot assay | 30365892 |
| MV4-11 | Function assay | 1000 nM | 6 hrs | Inhibition of HDAC1/2/3 in human MV4-11 cells assessed as upregulation of histone H3 acetylation at 1000 nM after 6 hrs by Western blot analysis | 26443078 |
| OPM2 | Cell Viability Assay | 0-8μM | 48 h | decreases MM-cell viability in a dose-dependent manner | 22262760 |
| MM.1R | Cell Viability Assay | 0-8μM | 48 h | decreases MM-cell viability in a dose-dependent manner | 22262760 |
| LR5 | Cell Viability Assay | 0-8μM | 48 h | decreases MM-cell viability in a dose-dependent manner | 22262760 |
| RPMI | Cell Viability Assay | 0-8μM | 48 h | decreases MM-cell viability in a dose-dependent manner | 22262760 |
| OPM1 | Cell Viability Assay | 0-8μM | 48 h | decreases MM-cell viability in a dose-dependent manner | 22262760 |
| MM.1S | Cell Viability Assay | 0-8μM | 48 h | decreases MM-cell viability in a dose-dependent manner | 22262760 |
| RPMI8226 | Function Assay | 0.25/1μM | 18 h | increases acetylated α-tubulin | 22262760 |
| MM.1S | Function Assay | 0.25/1μM | 18 h | increases acetylated α-tubulin | 22262760 |
| MM.1R | Function Assay | 0.25/1μM | 18 h | increases acetylated α-tubulin | 22262760 |
| MM.1S | Function Assay | 0-5μM | 6 h | increases acetylated α-tubulin | 22262760 |
| A-172 | Growth Inhibition Assay | 10 nM | 24/48 h | inhibits cell growth time dependently | 26150340 |
| U87MG | Growth Inhibition Assay | 10 nM | 24/48 h | inhibits cell growth time dependently | 26150340 |
| HEL | Cell cycle assay | 1 to 10 uM | 48 hrs | Cell cycle arrest in human HEL cells assessed as accumulation at G1 phase at 1 to 10 uM after 48 hrs propidium iodide staining based flow cytometry | 29940115 |
| SEM | Function assay | 1.6 uM | 18 hrs | Inhibition of HDAC6 in human SEM cells assessed as decrease in aggresome accumulation at 1.6 uM after 18 hrs by FACS analysis | 30365892 |
| SH-SY5Y | Function assay | 0.1 to 1 uM | 24 hrs | Inhibition of HDAC6 in human SH-SY5Y cells assessed as increase in acetylation of alpha-tubulin at 0.1 to 1 uM after 24 hrs by Western blot analysis | 30028616 |
| SH-SY5Y | Function assay | 0.1 to 1 uM | 24 hrs | Inhibition of class 1 HDAC in human SH-SY5Y cells assessed as increase in acetylation of histone H3 at 0.1 to 1 uM after 24 hrs by Western blot analysis | 30028616 |
| BCP-ALL | Cytotoxicity assay | 72 hrs | Cytotoxicity against human BCP-ALL cells derived from patient 3 after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 4.45 μM. | 30365892 | |
| HEL | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HEL cells after 48 hrs by CCK-8 assay, IC50 = 3.75 μM. | 29940115 | |
| K562 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human K562 cells after 48 hrs by CCK-8 assay, IC50 = 3.75 μM. | 29940115 | |
| HL60 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HL60 cells after 48 hrs by CCK-8 assay, IC50 = 3.75 μM. | 29940115 | |
| KCL22 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human KCL22 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 3.75 μM. | 30365892 | |
| SUP-B15 | Cytotoxicity assay | 72 hrs | Cytotoxicity against imatinib-resistant human SUP-B15 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 3.54 μM. | 30365892 | |
| U266 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human U266 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 3.52 μM. | 30365892 | |
| KCL22 | Cytotoxicity assay | 72 hrs | Cytotoxicity against imatinib-resistant human KCL22 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 3.38 μM. | 30365892 | |
| HEL | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HEL cells after 48 hrs in presence of JAK2 inhibitor CYT-387 by CCK-8 assay, IC50 = 2.54 μM. | 29940115 | |
| K562 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human K562 cells after 48 hrs in presence of JAK2 inhibitor CYT-387 by CCK-8 assay, IC50 = 2.54 μM. | 29940115 | |
| HL60 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HL60 cells after 48 hrs in presence of JAK2 inhibitor CYT-387 by CCK-8 assay, IC50 = 2.54 μM. | 29940115 | |
| HL60 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human HL60 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 2.36 μM. | 30365892 | |
| RPMI18226 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human RPMI18226 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 1.97 μM. | 30365892 | |
| SUP-B15 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SUP-B15 cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 1.92 μM. | 30365892 | |
| SEM | Cytotoxicity assay | 72 hrs | Cytotoxicity against human SEM cells after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 1.61 μM. | 30365892 | |
| RPMI8226 | Cytotoxicity assay | 72 hrs | Cytotoxicity against human RPMI8226 cells after 72 hrs by MTT assay, IC50 = 1.468 μM. | 26443078 | |
| BCP-ALL | Cytotoxicity assay | 72 hrs | Cytotoxicity against human BCP-ALL cells derived from patient 2 after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 0.58 μM. | 30365892 | |
| BCP-ALL | Cytotoxicity assay | 72 hrs | Cytotoxicity against human BCP-ALL cells derived from patient 4 after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 0.54 μM. | 30365892 | |
| BCP-ALL | Cytotoxicity assay | 72 hrs | Cytotoxicity against human BCP-ALL cells derived from patient 1 after 72 hrs by CellTiter-Glo luminescent cell viability assay, IC50 = 0.29 μM. | 30365892 | |
| Sf9 | Function assay | 15 mins | Inhibition of full length recombinant human C-terminal FLAG/His-tagged HDAC1 expressed in baculovirus infected sf9 cells using Boc-Lys-(Ac)-AMC as substrate preincubated for 15 mins followed by substrate addition measured after 60 mins by fluorescence ass, IC50 = 0.1 μM. | 29500130 | |
| Sf9 | Function assay | 15 mins | Inhibition of full length recombinant human C-terminal His-tagged HDAC2 expressed in baculovirus infected sf9 cells using Boc-Lys-(Ac)-AMC as substrate preincubated for 15 mins followed by substrate addition measured after 60 mins by fluorescence assay, IC50 = 0.066 μM. | 29500130 | |
| Sf9 | Function assay | 10 mins | Inhibition of full length human recombinant C-terminal FLAG-His-tagged HDAC1 expressed in Sf9 cells using FTS as substrate preincubated for 10 mins followed by substrate addition measured over 30 mins, IC50 = 0.058 μM. | 28038324 | |
| Sf9 | Function assay | 10 mins | Inhibition of full length human recombinant C-terminal FLAG-tagged HDAC2 expressed in Sf9 cells using FTS as substrate preincubated for 10 mins followed by substrate addition measured over 30 mins, IC50 = 0.048 μM. | 28038324 | |
| Sf9 | Function assay | 15 mins | Inhibition of full length recombinant human C-terminal His-tagged HDAC3/N-terminal GST-tagged NCOR2 (395 to 489 residues) expressed in baculovirus infected sf9 cells using Boc-Lys-(Ac)-AMC as substrate preincubated for 15 mins followed by substrate additi, IC50 = 0.037 μM. | 29500130 | |
| Sf9 | Function assay | 15 mins | Inhibition of full length recombinant human N-terminal GST-tagged HDAC6 expressed in baculovirus infected sf9 cells using Boc-Lys-(Ac)-AMC as substrate preincubated for 15 mins followed by substrate addition measured after 60 mins by fluorescence assay, IC50 = 0.009 μM. | 29500130 | |
| Sf9 | Function assay | 10 mins | Inhibition of full length human recombinant N-terminal GST-tagged HDAC6 expressed in Sf9 cells using FTS as substrate preincubated for 10 mins followed by substrate addition measured over 30 mins, IC50 = 0.0047 μM. | 28038324 | |
| HH | Growth Inhibition Assay | 48 h | IC50=2.5 μM | 26116270 | |
| Sup-T1 | Growth Inhibition Assay | 48 h | IC50=1.6 μM | 26116270 | |
| CCL-119 | Growth Inhibition Assay | 48 h | IC50=1.7 μM | 26116270 | |
| H9 | Growth Inhibition Assay | 48 h | IC50=1.2 μM | 26116270 | |
| Rec-1 | Growth Inhibition Assay | 48 h | IC50=2.3 μM | 26116270 | |
| Jeko-1 | Growth Inhibition Assay | 48 h | IC50=1.5 μM | 26116270 | |
| Jvm-2 | Growth Inhibition Assay | 48 h | IC50=4.0 μM | 26116270 | |
| Su-DHL6 | Growth Inhibition Assay | 48 h | IC50=3.2 μM | 26116270 | |
| Hbl-2 | Growth Inhibition Assay | 48 h | IC50=1.9 μM | 26116270 | |
| Su-DHL4 | Growth Inhibition Assay | 48 h | IC50=4.7 μM | 26116270 | |
| OCI-Ly1 | Growth Inhibition Assay | 48 h | IC50=2.4 μM | 26116270 | |
| OCI-Ly7 | Growth Inhibition Assay | 48 h | IC50=1.2 μM | 26116270 | |
| Su-DHL2 | Growth Inhibition Assay | 48 h | IC50=3.3 μM | 26116270 | |
| OCI-Ly10 | Growth Inhibition Assay | 48 h | IC50=0.9 μM | 26116270 | |
| Riva | Growth Inhibition Assay | 48 h | IC50=2.2 μM | 26116270 | |
| Hbl-1 | Growth Inhibition Assay | 48 h | IC50=1.6 μM | 26116270 | |
| SEM | Antiproliferative assay | 24 to 72 hrs | Antiproliferative activity against human SEM cells at IC50 to 2 times IC50 after 24 to 72 hrs by trypan exclusion method | 30365892 | |
| SEM | Function assay | 18 hrs | Inhibition of HDAC6 in human SEM cells assessed as decrease in aggresome accumulation at IC50 after 18 hrs by fluorescence microscopic method | 30365892 | |
| Rh41 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh41 cells | 29435139 | ||
| Rh30 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh30 cells | 29435139 | ||
| MG 63 (6-TG R) | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for MG 63 (6-TG R) cells | 29435139 | ||
| RD | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for RD cells | 29435139 | ||
| OHS-50 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for OHS-50 cells | 29435139 | ||
| Rh18 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Rh18 cells | 29435139 | ||
| BT-12 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-12 cells | 29435139 | ||
| LAN-5 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for LAN-5 cells | 29435139 | ||
| SK-N-SH | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-SH cells | 29435139 | ||
| NB1643 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB1643 cells | 29435139 | ||
| Saos-2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for Saos-2 cells | 29435139 | ||
| BT-37 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for BT-37 cells | 29435139 | ||
| NB-EBc1 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for NB-EBc1 cells | 29435139 | ||
| SK-N-MC | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SK-N-MC cells | 29435139 | ||
| A673 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for A673 cells | 29435139 | ||
| DAOY | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for DAOY cells | 29435139 | ||
| SJ-GBM2 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for SJ-GBM2 cells | 29435139 | ||
| TC32 | qHTS assay | qHTS of pediatric cancer cell lines to identify multiple opportunities for drug repurposing: Primary screen for TC32 cells | 29435139 | ||
| 他の多くの細胞株試験データをご覧になる場合はこちらをクリックして下さい | |||||
生物活性
| 製品説明 | Ricolinostat (ACY-1215, Rocilinostat) は、無細胞アッセイにおいて 5 nM の IC50 を持つ選択的 HDAC6 阻害剤です。HDAC1/2/3 (クラス I HDAC) よりも HDAC6 に対して 10 倍以上選択的で、HDAC8 に対してわずかな活性、HDAC4/5/7/9/11、Sirtuin1、Sirtuin2 に対しては最小限の活性しかありません。Ricolinostat (ACY-1215) は細胞増殖を抑制し、apoptosis を促進します。フェーズ 2。 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 特性 | Induced less cytotoxicity in PHA-stimulated PBMCs from 4 healthy donors compared with the pan-HDAC inhibitor SAHA. | ||||||||||
| Targets |
|
| In Vitro | ||||
| In vitro |
ACY-1215 is a hydroxamic acid derivative. ACY-1215 is 12-, 10-, and 11-fold less active against HDAC1, HDAC2, and HDAC3 (class I HDACs), respectively. ACY-1215 has minimal activity (IC50 > 1μM) against HDAC4, HDAC5, HDAC7, HDAC9, HDAC11, Sirtuin1, and Sirtuin2, and has slight activity against HDAC8 (IC50 = 0.1μM). The IC50 values for ACY-1215 for T-cell toxicity is 2.5μM. ACY-1215 overcomes tumor cell growth and survival conferred by BMSCs and cytokines in the BM milieu. |
|||
|---|---|---|---|---|
| Kinase Assay | HDAC enzymatic assays | |||
| ACY-1215 is dissolved and subsequently diluted in assay buffer [50 mM HEPES, pH 7.4, 100 mM KCl, 0.001% Tween-20, 0.05% BSA, and 20 μM tris(2-carboxyethyl)phosphine] to 6-fold the final concentration. HDAC enzymes are diluted to 1.5-fold of the final concentration in assay buffer and pre-incubated with ACY-1215 for 10 minutes before the addition of the substrate. The amount of FTS (HDAC1, HDAC2, HDAC3, and HDAC6) or MAZ-1675 (HDAC4, HDAC5, HDAC7, HDAC8, and HDAC9) used for each enzyme is equal to the Michaelis constant (Km), as determined by a titration curve. FTS or MAZ-1675 is diluted in assay buffer to 6-fold the final concentration with 0.3μM sequencing grade trypsin. The substrate/trypsin mix is added to the enzyme/compound mix and the plate is shaken for 60 seconds and then placed into a SpectraMax M5 microtiter plate reader. The enzymatic reaction is monitored for release of 7-amino-4-methoxy-coumarin over 30 minutes, after deacetylation of the lysine side chain in the peptide substrate, and the linear rate of the reaction is calculated. | ||||
| 細胞実験 | 細胞株 | MM cell lines, patient MM cells, and PBMCs | ||
| 濃度 | ~8 μM | |||
| 反応時間 | 48 hours | |||
| 実験の流れ | PBMCs from healthy donors are isolated and stimulated with 2.5 μg/mL of phytohemagglutinin (PHA) for 48 hours in the presence of increasing concentrations of ACY-1215. DNA synthesis is measured by tritiated thymidine uptake. CD4+T cells are purified from human blood with the Rosette Sep negative-selection kit. Cells are stimulated by CD3/CD28 Dynabeads for 7 days in the presence of compounds. |
|||
| 実験結果図 | Methods | Biomarkers | 結果図 | PMID |
| Western blot | Ac-α-tubulin / Ac-Histone H4 Survivin / P21 / CDC2 / p53 / p-p53(S392) / Cyclin A2 / Cyclin B1 Bax / Bim / Bcl2 / Cleaved caspase-3 / Cleaved caspase-9 / Cleaved PARP PI3K(p85) / AKT / p-AKT(S473) / PRAS40 / Rag C / mTOR / p-mTOR / ERK / p-ERK Ac-β-catenin(K49) / p-β-catenin / β-catenin |
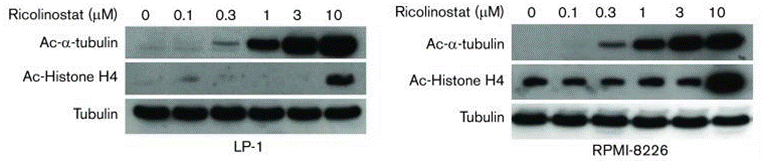
|
31015208 | |
| Immunofluorescence | β-tubulin / β-catenin |
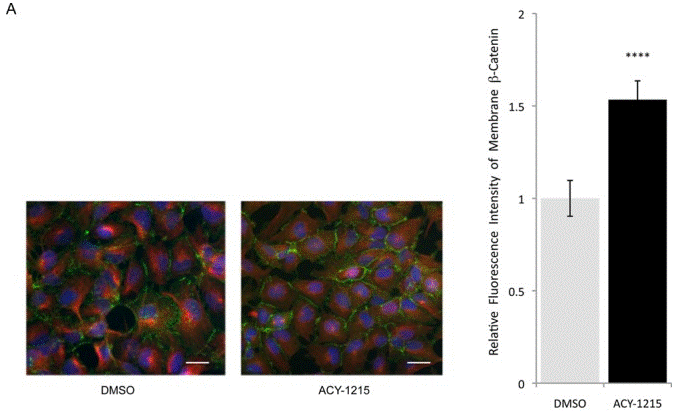
|
25546293 | |
| Growth inhibition assay | Cell viability |
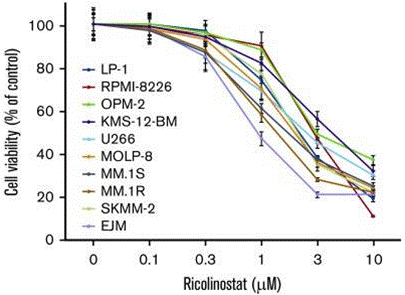
|
31015208 | |
| In Vivo | ||
| In Vivo |
ACY-1215 is readily absorbed by tumor tissue. Moreover, the drug does not accumulate in tumor tissue, as evidenced by the parallel decline of acetylated α-tubulin in blood cells and tumor tissue by 24 hours after dose. |
|
|---|---|---|
| 動物実験 | 動物モデル | MM xenograft SCID mouse model |
| 投与量 | 50 mg/kg | |
| 投与経路 | ip | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT02632071 | Completed | Metastatic Breast Cancer|Breast Carcinoma |
Columbia University|Acetylon Pharmaceuticals Incorporated|National Cancer Institute (NCI) |
March 1 2016 | Phase 1 |
| NCT01583283 | Completed | Multiple Myeloma |
Celgene |
July 12 2012 | Phase 1 |
| NCT01323751 | Completed | Multiple Myeloma |
Celgene|The Leukemia and Lymphoma Society |
July 2011 | Phase 1|Phase 2 |
|
化学情報
| 分子量 | 433.5 | 化学式 | C24H27N5O3 |
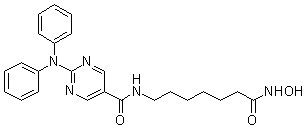
| CAS No. | 1316214-52-4 | SDF | Download Ricolinostat (ACY-1215) SDFをダウンロードする |
| Smiles | C1=CC=C(C=C1)N(C2=CC=CC=C2)C3=NC=C(C=N3)C(=O)NCCCCCCC(=O)NO | ||
| 保管 | |||
|
In vitro |
DMSO : 42 mg/mL ( (96.88 mM); 吸湿したDMSOは溶解度を減少させます。新しいDMSOをご使用ください。) Water : Insoluble Ethanol : Insoluble |
モル濃度計算器 |
|
in vivo Add solvents to the product individually and in order. |
投与溶液組成計算機 | |||||
実験計算
投与溶液組成計算機(クリア溶液)
ステップ1:実験データを入力してください。(実験操作によるロスを考慮し、動物数を1匹分多くして計算・調製することを推奨します)
mg/kg
g
μL
匹
ステップ2:投与溶媒の組成を入力してください。(ロット毎に適した溶解組成が異なる場合があります。詳細については弊社までお問い合わせください)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
計算結果:
投与溶媒濃度: mg/ml;
DMSOストック溶液調製方法: mg 試薬を μL DMSOに溶解する(濃度 mg/mL, 注:濃度が当該ロットのDMSO溶解度を超える場合はご連絡ください。 )
投与溶媒調製方法:Take μL DMSOストック溶液に μL PEG300,を加え、完全溶解後μL Tween 80,を加えて完全溶解させた後 μL ddH2O,を加え完全に溶解させます。
投与溶媒調製方法:μL DMSOストック溶液に μL Corn oil,を加え、完全溶解。
注意:1.ストック溶液に沈殿、混濁などがないことをご確認ください;
2.順番通りに溶剤を加えてください。次のステップに進む前に溶液に沈殿、混濁などがないことを確認してから加えてください。ボルテックス、ソニケーション、水浴加熱など物理的な方法で溶解を早めることは可能です。
技術サポート
ストックの作り方、阻害剤の保管方法、細胞実験や動物実験の際に注意すべき点など、製品を取扱う時に問い合わせが多かった質問に対しては取扱説明書でお答えしています。
他に質問がある場合は、お気軽にお問い合わせください。
* 必須
よくある質問(FAQ)
質問1:
What would you suggest to obtain a clear solution of it?
回答
It can be dissolved in 2% DMSO/30% PEG 300/ddH2O at 5 mg/ml clearly, while in 1% DMSO/30% polyethylene glycol/1% Tween 80 at 30 mg/ml it is a suspension for oral administration. Please note that the precipitation will go out from the clear solution after stayed for about half an hour, so it is recommended to prepare the solution just before use.