- 阻害剤
- 研究分野別
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- 化合物ライブラリー
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- Customize Library(compound antibody kit)
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-II
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleck.co.jp to customize your library.
You could select:
- 抗体
- 新製品
- お問い合わせ
Dacomitinib (PF-299804)
別名:PF299804,PF299
Dacomitinibは、主にEGFRに対して6nMのIC50を示す強力な不可逆的パン-ErbB阻害剤であり、セルフリーアッセイで確認されています。DacomitinibはERBB2とERBB4をそれぞれ45.7nMと73.7nMのIC50で阻害します。Dacomitinibは、EGFRまたはERBB2変異(ゲフィチニブ耐性)を持つNSCLC、およびEGFR T790M変異を持つNSCLCに対しても効果的です。Dacomitinibは細胞増殖を抑制し、apoptosisを誘導します。第2相。
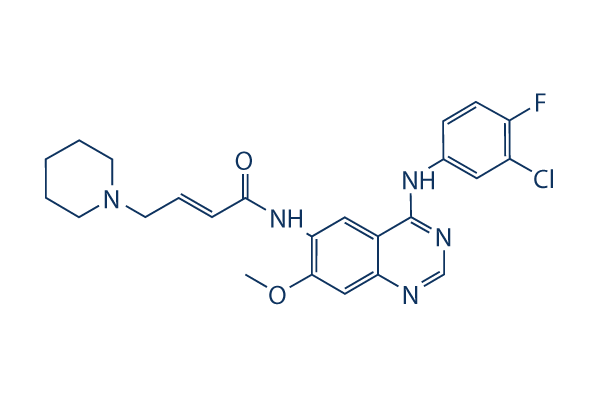
CAS No. 1110813-31-4
文献中Selleckの製品使用例(96)
製品安全説明書
現在のバッチを見る:
純度:
99.81%
99.81
Dacomitinib (PF-299804)関連製品
シグナル伝達経路
EGFR阻害剤の選択性比較
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | 活性情報 | PMID |
|---|---|---|---|---|---|
| NIH/3T3 | Function assay | 30 mg/kg | 2 days | In vivo inhibition of full length human ERBB1 autophosphorylation transfected in NIH/3T3 cells implanted in mouse at 30 mg/kg, po qd for 2 days measured 24 hrs post last dose by Western blot analysis | 27491023 |
| human NCI-H1975 cells | Function assay | 2 h | Inhibition of EGFR L858R/T970M double mutant phosphorylation in human NCI-H1975 cells after 2 hrs by fluorescence assay, IC50=0.042 μM | 23930994 | |
| human LoVo cells | Function assay | 2 h | Inhibition of wild type EGFR phosphorylation in human LoVo cells after 2 hrs by fluorescence assay, IC50=0.011 μM | 23930994 | |
| PC9 cells | Function assay | 2 h | Inhibition of EGFR exon 19 deletion activating mutant phosphorylation in human PC9 cells after 2 hrs by fluorescence assay, IC50=0.63 nM | 23930994 | |
| Sf9 | Function assay | 6 mins | Irreversible inhibition of GST-tagged ERBB1 (unknown origin) (Met-668 to Ala-1211 residues) expressed in baculovirus infected Sf9 insect cells assessed as reduction in Glu/Tyr copolymer phosphorylation after 6 mins by ELISA, IC50 = 0.006 μM. | 27491023 | |
| NIH/3T3 | Function assay | 2 hrs | Irreversible inhibition of full length human ERBB1 autophosphorylation transfected in EGF-stimulated mouse NIH/3T3 cells incubated for 2 hrs followed by stimulation with EGF for 10 mins, IC50 = 0.006 μM. | 27491023 | |
| Sf9 | Function assay | 6 mins | Irreversible inhibition of GST-tagged ERBB2 (unknown origin) (Ile-675 to Val-1256 residues) expressed in baculovirus infected Sf9 insect cells assessed as reduction in Glu/Tyr copolymer phosphorylation after 6 mins by ELISA, IC50 = 0.046 μM. | 27491023 | |
| Sf9 | Function assay | 6 mins | Irreversible inhibition of GST-tagged ERBB4 (unknown origin) (Gly-259 to Gly-690 residues) expressed in baculovirus infected Sf9 insect cells assessed as reduction in Glu/Tyr copolymer phosphorylation after 6 mins by ELISA, IC50 = 0.074 μM. | 27491023 | |
| Sf9 | Function assay | 30 mins | Irreversible inhibition of human recombinant GST-tagged JAK3 expressed in baculovirus infected Sf9 insect cells assessed as reduction in polyglutamic acid-tyrosine phosphorylation after 30 mins by ELISA, IC50 = 3.57 μM. | 27491023 | |
| NCI-H1819 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human NCI-H1819 cells expressing wild type HER2 incubated for 72 hrs by MTS assay, IC50 = 0.029 μM. | 28754471 | |
| NCI-H1975 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human NCI-H1975 cells expressing EGFR T790M/L858R mutant incubated for 72 hrs by MTS assay, IC50 = 0.44 μM. | 28754471 | |
| human NCI-H1975 cells | Proliferation assay | Antiproliferative activity against human NCI-H1975 cells assessed as growth inhibition, GI50=0.1233 μM | 26310890 | ||
| insect cells | Function assay | Inhibition of GST-tagged human EGFR catalytic domain expressed in insect cells, IC50 = 0.006 μM. | 28754471 | ||
| insect cells | Function assay | Inhibition of GST-tagged human HER2 catalytic domain expressed in insect cells, IC50 = 0.0457 μM. | 28754471 | ||
| 他の多くの細胞株試験データをご覧になる場合はこちらをクリックして下さい | |||||
生物活性
| 製品説明 | Dacomitinibは、主にEGFRに対して6nMのIC50を示す強力な不可逆的パン-ErbB阻害剤であり、セルフリーアッセイで確認されています。DacomitinibはERBB2とERBB4をそれぞれ45.7nMと73.7nMのIC50で阻害します。Dacomitinibは、EGFRまたはERBB2変異(ゲフィチニブ耐性)を持つNSCLC、およびEGFR T790M変異を持つNSCLCに対しても効果的です。Dacomitinibは細胞増殖を抑制し、apoptosisを誘導します。第2相。 | ||||||
|---|---|---|---|---|---|---|---|
| Targets |
|
| In Vitro | ||||
| In vitro |
PF299804 is a specific inhibitor of the ERBB family of kinases. This compound inhibits EGFR signaling and induces apoptosis in the EGFR T790M-containing H3255 GR cell line. It is effective in -sensitive and NSCLC cell lines. This chemical inhibits the growth of H3255 and HCC827 cells engineered to express EGFR T790M. It inhibits EGFR phosphorylation in the presence of the T790M mutation. This agent is believed to irreversibly inhibit ERBB tyrosine kinase activity through binding at the ATP site and covalent modification of nucleophilic cysteine residues in the catalytic domains of ERBB family members. It shows significant growth-inhibitory effects in HER2-amplified gastric cancer cells (SNU216, N87), and it has lower 50% inhibitory concentration values compared with other EGFR tyrosine kinase inhibitors, including BIBW-2992, and CI-1033. This inhibitor induces apoptosis and G1 arrest and inhibits phosphorylation of receptors in the HER family and downstream signaling pathways including STAT3, AKT, and extracellular signal-regulated kinases (ERK) in HER2-amplified gastric cancer cells. It also blocks EGFR/HER2, HER2/HER3, and HER3/HER4 heterodimer formation as well as the association of HER3 with p85α in SNU216 cells. A recent research uses forty-seven human breast cancer and immortalized breast epithelial lines to evaluate the inhibition effects of this compound, the results indicate it preferentially inhibits growth of HER-2-amplified breast cancer cell lines than nonamplified lines (RR = 3.39, p < 0.0001). This chemical reduces the phosphorylation of HER2, EGFR, HER4, AKT, and ERK in the majority of sensitive lines. It exerts its anti-proliferative effect through a combined G0/G1 arrest and an induction of apoptosis. |
|||
|---|---|---|---|---|
| Kinase Assay | ELISA-Based ERBB Kinase Assay | |||
| The ERBB1, ERBB 2, and ERBB4 cytoplasmic fusion proteins are made by cloning the ERBB1 sequence (Met-668 to Ala-1211), ERBB2 (Ile-675 to Val-1256), and ERBB4 sequence (Gly-259 to Gly-690) into the baculoviral vector pFastBac using PCR. Proteins are expressed in baculovirusinfected Sf9 insect cells as GST fusion proteins. The proteins are purified by affinity chromatography using glutathione sepharose beads. Inhibition of ERBB tyrosine kinase activity is assessed using an ELISA-based receptor tyrosine kinase assay. Kinase reactions (50 mM HEPES, pH 7.4, 125 mM NaCl, 10 mM MgCl2, 100 μM sodium orthovanadate, 2 mM dithiothreitol, 20 μM ATP, this compound or vehicle control, and 1-5 nM GST-erbB per 50 μL of reaction mixture) are run in 96-well plates coated with 0.25 mg/mL poly-Glu-Tyr. The reactions are incubated for 6 minutes at room temperature while being shaken. Kinase reactions are stopped by removal of the reaction mixture, and then the wells are washed with wash buffer (0.1% Tween 20 in PBS). Phosphorylated tyrosine residues are detected by adding 0.2 μg/mL antiphosphotyrosine antibody (Oncogene Ab-4; 50 μL/well) coupled to horseradish peroxidase (HRP) diluted in PBS containing 3% BSA and 0.05% Tween 20 for 25 minutes while being shaken at room temperature. The antibody is removed, and plates are washed in wash buffer. HRP substrate (SureBlue3,3,5,5-tetramethyl benzidine or TMB) is added (50 μL per well) and incubated for 10-20 minutes while it is shaken at room temperature. The TMB reaction is stopped with the addition of 50 μL of stop solution (0.09 N H2SO4). The signal is quantified by measuring absorbance at 450 nm. IC50 values are determined for this compound using the median effect method. | ||||
| 細胞実験 | 細胞株 | Various NSCLC cell lines | ||
| 濃度 | 0-20 nM | |||
| 反応時間 | 72 hours | |||
| 実験の流れ | Growth and inhibition of growth is assessed by 5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS) assay. This assay, a colorimetric method fordetermining the number of viable cells, is based on the bioreduction of MTS by cells to a formazan product that is soluble in cell culture medium, can be detected spectrophotometrically. The cells are exposed totreatment for 72 hours, and the number of cells used per experiment is determined empirically. All experimental points are set up in 6 to 12 wells, and all experiments are repeated at least thrice. The data are graphically displayed using GraphPad Prism version 3.00 for Windows (GraphPad Software). The curves are fitted using a nonlinear regression model with a sigmoidal dose response. |
|||
| 実験結果図 | Methods | Biomarkers | 結果図 | PMID |
| Western blot | pEGFR / EGFR / pERK / ERK / pAKT / AKT / p-mTOR / mTOR / pSTAT3 / STAT3 |
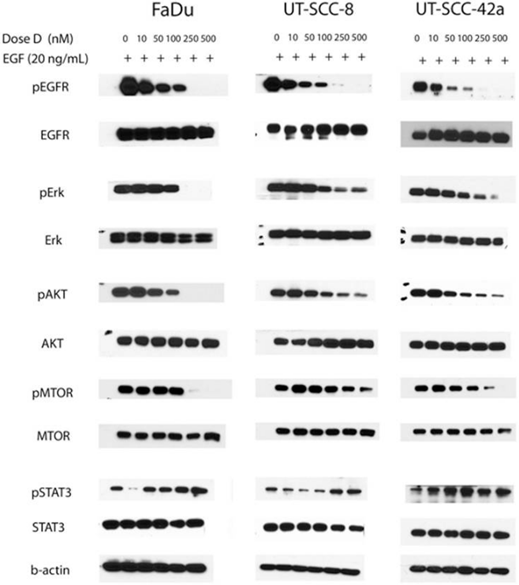
|
24853121 | |
| Immunofluorescence | LC3 |
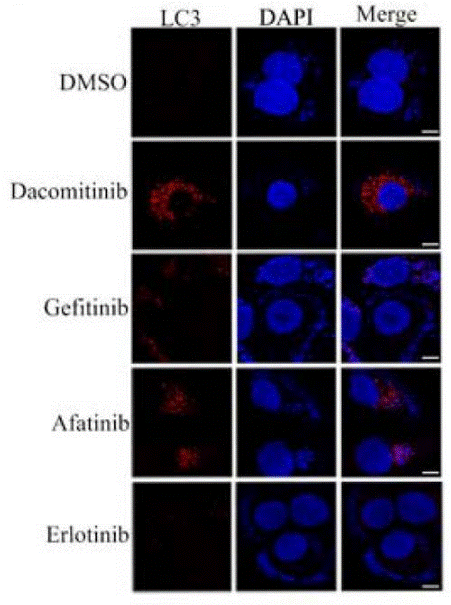
|
28366635 | |
| Growth inhibition assay | Cell viability |
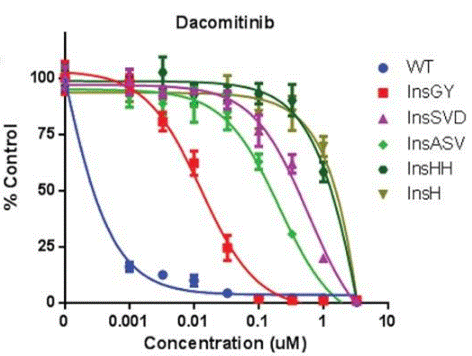
|
28363995 | |
| In Vivo | ||
| In Vivo |
Orally administered PF299804 effectively inhibits growth of HCC827 Del/T790M xenografts. Low oral administration of this compound (15mg/kg) causes significant antitumor activity, including marked tumor regressions in a variety of human tumor xenograft models that express and/ or overexpress ERBB family members or contain the double mutation (L858R/T790M) in ERBB1 (EGFR). |
|
|---|---|---|
| 動物実験 | 動物モデル | HCC827-GFP or HCC827-Del/T790M lung cancer cells (in 0.2 mL of PBS) are inoculated s.c. into the lower-right quadrant of the flank of nude mice |
| 投与量 | 10 mg/kg | |
| 投与経路 | Administered via oral gavage | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06075615 | Not yet recruiting | Carcinoma Non-Small-Cell Lung |
Pfizer |
September 1 2024 | -- |
| NCT06321510 | Not yet recruiting | Lung Cancer |
Pfizer |
April 1 2024 | -- |
| NCT04609319 | Active not recruiting | Lung Cancer |
Pfizer |
January 23 2021 | -- |
| NCT03865446 | Completed | Severe Hepatic Impairment |
Pfizer |
April 5 2019 | Phase 1 |
| NCT02382796 | Completed | NSCLC |
Pfizer |
July 10 2015 | Phase 2 |
| NCT02268747 | Unknown status | Skin Squamous Cell Cancer |
Fondazione IRCCS Istituto Nazionale dei Tumori Milano |
November 2014 | Phase 2 |
|
化学情報
| 分子量 | 469.94 | 化学式 | C24H25ClFN5O2 |
| CAS No. | 1110813-31-4 | SDF | Download Dacomitinib (PF-299804) SDFをダウンロードする |
| Smiles | COC1=C(C=C2C(=C1)N=CN=C2NC3=CC(=C(C=C3)F)Cl)NC(=O)C=CCN4CCCCC4 | ||
| 保管 | |||
|
In vitro |
DMSO : 12.5 mg/mL ( (26.59 mM); 吸湿したDMSOは溶解度を減少させます。新しいDMSOをご使用ください。) Water : Insoluble Ethanol : Insoluble |
モル濃度計算器 |
|
in vivo Add solvents to the product individually and in order. |
投与溶液組成計算機 | |||||
実験計算
投与溶液組成計算機(クリア溶液)
ステップ1:実験データを入力してください。(実験操作によるロスを考慮し、動物数を1匹分多くして計算・調製することを推奨します)
mg/kg
g
μL
匹
ステップ2:投与溶媒の組成を入力してください。(ロット毎に適した溶解組成が異なる場合があります。詳細については弊社までお問い合わせください)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
計算結果:
投与溶媒濃度: mg/ml;
DMSOストック溶液調製方法: mg 試薬を μL DMSOに溶解する(濃度 mg/mL, 注:濃度が当該ロットのDMSO溶解度を超える場合はご連絡ください。 )
投与溶媒調製方法:Take μL DMSOストック溶液に μL PEG300,を加え、完全溶解後μL Tween 80,を加えて完全溶解させた後 μL ddH2O,を加え完全に溶解させます。
投与溶媒調製方法:μL DMSOストック溶液に μL Corn oil,を加え、完全溶解。
注意:1.ストック溶液に沈殿、混濁などがないことをご確認ください;
2.順番通りに溶剤を加えてください。次のステップに進む前に溶液に沈殿、混濁などがないことを確認してから加えてください。ボルテックス、ソニケーション、水浴加熱など物理的な方法で溶解を早めることは可能です。
技術サポート
ストックの作り方、阻害剤の保管方法、細胞実験や動物実験の際に注意すべき点など、製品を取扱う時に問い合わせが多かった質問に対しては取扱説明書でお答えしています。
他に質問がある場合は、お気軽にお問い合わせください。
* 必須
よくある質問(FAQ)
質問1:
I would like to know whether the in vivo formulation you recommend for it is suitable for oral administration?
回答
S2727 in 1% DMSO+30% polyethylene glycol+1% Tween 80 at 10mg/ml is a homogeneous suspension, and it was fine for oral gavage. When preparing the solution, please dissolve it in DMSO clearly first. If it dissolves not readily, please sonicate and warm it at about 45-50℃ for a while to help dissolving. Then add PEG 300 and Tween 80. After they mixed well, dilute with water. Then it will become a homogeneous suspension.