- 阻害剤
- 研究分野別
- PI3K/Akt/mTOR
- Epigenetics
- Methylation
- Immunology & Inflammation
- Protein Tyrosine Kinase
- Angiogenesis
- Apoptosis
- Autophagy
- ER stress & UPR
- JAK/STAT
- MAPK
- Cytoskeletal Signaling
- Cell Cycle
- TGF-beta/Smad
- 化合物ライブラリー
- Popular Compound Libraries
- Customize Library
- Clinical and FDA-approved Related
- Bioactive Compound Libraries
- Inhibitor Related
- Natural Product Related
- Metabolism Related
- Cell Death Related
- By Signaling Pathway
- By Disease
- Anti-infection and Antiviral Related
- Neuronal and Immunology Related
- Fragment and Covalent Related
- Customize Library(compound antibody kit)
- FDA-approved Drug Library
- FDA-approved & Passed Phase I Drug Library
- Preclinical/Clinical Compound Library
- Bioactive Compound Library-I
- Bioactive Compound Library-II
- Kinase Inhibitor Library
- Express-Pick Library
- Natural Product Library
- Human Endogenous Metabolite Compound Library
- Alkaloid Compound LibraryNew
- Angiogenesis Related compound Library
- Anti-Aging Compound Library
- Anti-alzheimer Disease Compound Library
- Antibiotics compound Library
- Anti-cancer Compound Library
- Anti-cancer Compound Library-Ⅱ
- Anti-cancer Metabolism Compound Library
- Anti-Cardiovascular Disease Compound Library
- Anti-diabetic Compound Library
- Anti-infection Compound Library
- Antioxidant Compound Library
- Anti-parasitic Compound Library
- Antiviral Compound Library
- Apoptosis Compound Library
- Autophagy Compound Library
- Calcium Channel Blocker LibraryNew
- Cambridge Cancer Compound Library
- Carbohydrate Metabolism Compound LibraryNew
- Cell Cycle compound library
- CNS-Penetrant Compound Library
- Covalent Inhibitor Library
- Cytokine Inhibitor LibraryNew
- Cytoskeletal Signaling Pathway Compound Library
- DNA Damage/DNA Repair compound Library
- Drug-like Compound Library
- Endoplasmic Reticulum Stress Compound Library
- Epigenetics Compound Library
- Exosome Secretion Related Compound LibraryNew
- FDA-approved Anticancer Drug LibraryNew
- Ferroptosis Compound Library
- Flavonoid Compound Library
- Fragment Library
- Glutamine Metabolism Compound Library
- Glycolysis Compound Library
- GPCR Compound Library
- Gut Microbial Metabolite Library
- HIF-1 Signaling Pathway Compound Library
- Highly Selective Inhibitor Library
- Histone modification compound library
- HTS Library for Drug Discovery
- Human Hormone Related Compound LibraryNew
- Human Transcription Factor Compound LibraryNew
- Immunology/Inflammation Compound Library
- Inhibitor Library
- Ion Channel Ligand Library
- JAK/STAT compound library
- Lipid Metabolism Compound LibraryNew
- Macrocyclic Compound Library
- MAPK Inhibitor Library
- Medicine Food Homology Compound Library
- Metabolism Compound Library
- Methylation Compound Library
- Mouse Metabolite Compound LibraryNew
- Natural Organic Compound Library
- Neuronal Signaling Compound Library
- NF-κB Signaling Compound Library
- Nucleoside Analogue Library
- Obesity Compound Library
- Oxidative Stress Compound LibraryNew
- Phenotypic Screening Library
- PI3K/Akt Inhibitor Library
- Protease Inhibitor Library
- Protein-protein Interaction Inhibitor Library
- Pyroptosis Compound Library
- Small Molecule Immuno-Oncology Compound Library
- Mitochondria-Targeted Compound LibraryNew
- Stem Cell Differentiation Compound LibraryNew
- Stem Cell Signaling Compound Library
- Natural Phenol Compound LibraryNew
- Natural Terpenoid Compound LibraryNew
- TGF-beta/Smad compound library
- Traditional Chinese Medicine Library
- Tyrosine Kinase Inhibitor Library
- Ubiquitination Compound Library
-
Cherry Picking
You can personalize your library with chemicals from within Selleck's inventory. Build the right library for your research endeavors by choosing from compounds in all of our available libraries.
Please contact us at info@selleck.co.jp to customize your library.
You could select:
- 抗体
- 新製品
- お問い合わせ
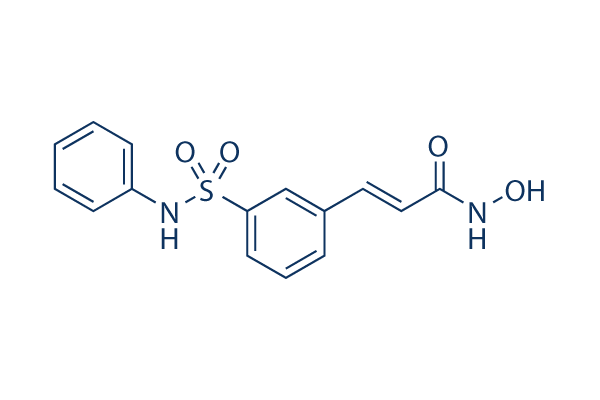
Belinostat (PXD101)
別名:PXD101,NSC726630, PX-105684
Belinostatは、細胞フリーアッセイで27 nMのIC50を持つ新規のHDAC阻害剤であり、シスプラチン耐性腫瘍で活性が実証されています。Belinostat(PXD101)はautophagyを誘導します。
CAS No. 866323-14-0
文献中Selleckの製品使用例(106)
製品安全説明書
現在のバッチを見る:
純度:
99.74%
99.74
Belinostat (PXD101)関連製品
シグナル伝達経路
HDAC阻害剤の選択性比較
Cell Data
| Cell Lines | Assay Type | Concentration | Incubation Time | 活性情報 | PMID |
|---|---|---|---|---|---|
| PC9 | Function Assay | 500 nM | 24 h | decreases EGFR expression | 23515752 |
| H460 | Function Assay | 0.5/1/2 μM | 4 h | inhibits the levels of Akt (p-Akt) and EGFR | 23515752 |
| H1650 | Function Assay | 0.5/1/2 μM | 4 h | inhibits the levels of Akt (p-Akt) and EGFR | 23515752 |
| PC9 | Function Assay | 0.5/1/2 μM | 4 h | inhibits the levels of Akt (p-Akt) and EGFR | 23515752 |
| Panc0327 | Apoptosis Assay | 1 μM | 24 h | induces apoptosis | 23475695 |
| Panc1005 | Apoptosis Assay | 1 μM | 24 h | induces apoptosis | 23475695 |
| Panc0403 | Apoptosis Assay | 1 μM | 24 h | induces apoptosis | 23475695 |
| AsPc1 | Function Assay | 1/10 μM | 24 h | induces growth arrested in G2/M | 23475695 |
| MiaPaCa2 | Function Assay | 1/10 μM | 24 h | induces growth arrested in G2/M | 23475695 |
| T3M4 | Growth Inhibition Assay | 0-800 nM | 48 h | inhibits cell proliferation in a dose dependent manner | 22681698 |
| AsPC-1 | Growth Inhibition Assay | 0-800 nM | 48 h | inhibits cell proliferation in a dose dependent manner | 22681698 |
| Panc-1 | Growth Inhibition Assay | 0-800 nM | 48 h | inhibits cell proliferation in a dose dependent manner | 22681698 |
| T3M4 | Apoptosis Assay | 100/500/1000 nM | 48 h | induces dose dependent apoptosis | 22681698 |
| AsPC-1 | Apoptosis Assay | 100/500/1000 nM | 48 h | induces dose dependent apoptosis | 22681698 |
| Panc-1 | Apoptosis Assay | 100/500/1000 nM | 48 h | induces dose dependent apoptosis | 22681698 |
| HCT116 | Function Assay | 0.9 μM | 24 h | down-regulats TS protein levels after 6 h incubation | 17124594 |
| H1650 | Function Assay | 500 nM | 24 h | decreases EGFR expression | 23515752 |
| H460 | Function Assay | 500 nM | 24 h | decreases EGFR expression | 23515752 |
| PANC-1 | Function Assay | 10 μM | 2/4 h | increases intracellular ROS level | 23743198 |
| PANC-1 | Cell Viability Assay | 1/10 μM | 48 h | decreases cell viability in a dose dependent manner | 23743198 |
| PANC-1 | Function Assay | 10 μM | 2/4/6 h | induces AMPK activation | 23743198 |
| HL-60 | Function Assay | 0.2 μM | 24/48/72 h | enhances RA-induced granulocytic differentiation | 25864732 |
| NB4 | Function Assay | 0.2 μM | 24/48/72 h | enhances RA-induced granulocytic differentiation | 25864732 |
| HL-60 | Function Assay | 2 μM | 24/48 h | blocks cell cycle in S phase | 25864732 |
| NB4 | Function Assay | 2 μM | 24/48 h | blocks cell cycle in S phase | 25864732 |
| HL-60 | Cell Viability Assay | 0.2/2 μM | 24/48/72 h | decreases cell viability in both time and dose dependent manner | 25864732 |
| NB4 | Cell Viability Assay | 0.2/2 μM | 24/48/72 h | decreases cell viability in both time and dose dependent manner | 25864732 |
| Huh-luc/neo7 | Function assay | 1 uM | 1 to 3 hrs | Inhibition of HDAC class 1 in human Huh-luc/neo7 cells assessed as histone H3 acetylation at 1 uM after 1 to 3 hrs by Western blotting analysis | 25937017 |
| PC3 | Function assay | 0.3 uM | 48 hrs | Inhibition of HDAC in human PC3 cells assessed as increase in amount of acetylated histone H3 at 0.3 uM after 48 hrs by Western blot analysis | 27344487 |
| HCT116 | Function assay | 0.3 uM | 48 hrs | Inhibition of HDAC in human HCT116 cells assessed as increase in amount of acetylated histone H3 at 0.3 uM after 48 hrs by Western blot analysis | 27344487 |
| AsPc1 | Growth Inhibition Assay | 48 h | EC50=0.3 μM | 23475695 | |
| Panc0327 | Growth Inhibition Assay | 48 h | EC50=0.5 μM | 23475695 | |
| MiaPaCa2 | Growth Inhibition Assay | 48 h | EC50=0.7 μM | 23475695 | |
| BxPc3 | Growth Inhibition Assay | 48 h | EC50=1.0 μM | 23475695 | |
| Panc0403 | Growth Inhibition Assay | 48 h | EC50=1.1 μM | 23475695 | |
| Panc1005 | Growth Inhibition Assay | 48 h | EC50=1.1 μM | 23475695 | |
| PL45 | Growth Inhibition Assay | 48 h | EC50=20.8 μM | 23475695 | |
| Panc0203 | Growth Inhibition Assay | 48 h | EC50=22.2 μM | 23475695 | |
| HBL-2 | Growth Inhibition Assay | 24 h | IC50=0.4 μM | 20068080 | |
| Jeko-1 | Growth Inhibition Assay | 24 h | IC50=0.2 μM | 20068080 | |
| Granta-519 | Growth Inhibition Assay | 24 h | IC50=56.3 μM | 20068080 | |
| HCT116 | Growth Inhibition Assay | 48 h | EC50=0.28 μM | 17124594 | |
| HCC4006 | Growth Inhibition Assay | 72 h | IC50=0.46 μM | 23515752 | |
| HCC2935 | Growth Inhibition Assay | 72 h | IC50=0.97 μM | 23515752 | |
| HCC827 | Growth Inhibition Assay | 72 h | IC50=0.29 μM | 23515752 | |
| HCC2279 | Growth Inhibition Assay | 72 h | IC50=0.4 μM | 23515752 | |
| PC9 | Growth Inhibition Assay | 72 h | IC50=0.29 μM | 23515752 | |
| H820 | Growth Inhibition Assay | 72 h | IC50=0.4 μM | 23515752 | |
| H1650 | Growth Inhibition Assay | 72 h | IC50=0.88 μM | 23515752 | |
| H1975 | Growth Inhibition Assay | 72 h | IC50=0.68 μM | 23515752 | |
| H520 | Growth Inhibition Assay | 72 h | IC50=0.75 μM | 23515752 | |
| H1299 | Growth Inhibition Assay | 72 h | IC50=1.2 μM | 23515752 | |
| H460 | Growth Inhibition Assay | 72 h | IC50=0.86 μM | 23515752 | |
| H1666 | Growth Inhibition Assay | 72 h | IC50>10 μM | 23515752 | |
| RAW264.7 | Anti-inflammatory assay | 1 hr | Anti-inflammatory activity in LPS-stimulated mouse RAW264.7 cells assessed as suppression of IL6 production pre-incubated for 1 hr before LPS stimulation for 24 hrs by ELISA method, IC50 = 0.000059 μM. | 25113875 | |
| HeLa | Function assay | 30 mins | Inhibition of HDAC in human HeLa cells nuclear extracts incubated for 30 mins by fluorescent assay, IC50 = 0.0264 μM. | 25113875 | |
| MDA-MB-231 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MDA-MB-231 cells after 72 hrs by resazurin dye based fluorescence assay, IC50 = 0.062 μM. | 29456804 | |
| Jurkat | Antiproliferative assay | 48 hrs | Antiproliferative activity against human Jurkat cells after 48 hrs by MTT assay, IC50 = 0.07 μM. | 29533873 | |
| A549 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human A549 cells after 72 hrs by resazurin dye based fluorescence assay, IC50 = 0.077 μM. | 29456804 | |
| HeLa | Antiproliferative assay | 72 hrs | Antiproliferative activity against human HeLa cells after 72 hrs by resazurin dye based fluorescence assay, IC50 = 0.087 μM. | 29456804 | |
| MCF7 | Antiproliferative assay | 72 hrs | Antiproliferative activity against human MCF7 cells after 72 hrs by resazurin dye based fluorescence assay, IC50 = 0.096 μM. | 29456804 | |
| HEL | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HEL cells after 48 hrs by MTT assay, IC50 = 0.1 μM. | 29533873 | |
| Huh7 | Antiviral assay | 3 days | Antiviral activity against HCV genotype 1b infected in human Huh7 cells after 3 days by luciferase reporter gene assay, EC50 = 0.12 μM. | 25490700 | |
| HCT116 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HCT116 cells after 48 hrs by SRB assay, GI50 = 0.13 μM. | 27344487 | |
| MOLT4 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human MOLT4 cells after 48 hrs by MTT assay, IC50 = 0.14 μM. | 29533873 | |
| SK-N-BE(2) | Antiproliferative assay | 48 hrs | Antiproliferative activity against human SK-N-BE(2) cells after 48 hrs by MTT assay, IC50 = 0.31 μM. | 29533873 | |
| PC3 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human PC3 cells after 48 hrs by SRB assay, GI50 = 0.39 μM. | 27344487 | |
| PC3 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human PC3 cells after 96 hrs by celltiter 96 assay, IC50 = 0.45 μM. | 21634430 | |
| HeLa | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HeLa cells after 48 hrs by MTT assay, IC50 = 0.51 μM. | 29533873 | |
| HCT116 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human HCT116 cells after 96 hrs by celltiter 96 assay, IC50 = 0.6 μM. | 21634430 | |
| A2780 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human A2780 cells after 96 hrs by celltiter 96 assay, IC50 = 0.67 μM. | 21634430 | |
| HuH7 | Cytotoxicity assay | 3 days | Cytotoxicity against human HuH7 cells assessed as inhibition of cell viability after 3 days by CellTiter 96 assay, CC50 = 0.68 μM. | 25490700 | |
| COLO205 | Antiproliferative assay | 96 hrs | Antiproliferative activity against human COLO205 cells after 96 hrs by celltiter 96 assay, IC50 = 0.7 μM. | 21634430 | |
| A549 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human A549 cells after 48 hrs by SRB assay, GI50 = 0.78 μM. | 27344487 | |
| HL60 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human HL60 cells after 48 hrs by SRB assay, GI50 = 1.09 μM. | 27344487 | |
| K562 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human K562 cells after 48 hrs by MTT assay, IC50 = 1.1 μM. | 29533873 | |
| PC3 | Antiproliferative assay | 48 hrs | Antiproliferative activity against human PC3 cells after 48 hrs by MTT assay, IC50 = 1.3 μM. | 29533873 | |
| NFF | Cytotoxicity assay | 72 hrs | Cytotoxicity against human NFF cells after 72 hrs by SRB assay, IC50 = 1.4 μM. | 28241112 | |
| HEK293 | Cytotoxicity assay | 48 hrs | Cytotoxicity against HEK293 cells after 48 hrs by resazurin assay, IC50 = 1.4 μM. | 28241112 | |
| NFF | Cytotoxicity assay | 72 hrs | Cytotoxicity against human NFF cells after 72 hrs by sulforhodamine B assay, IC50 = 1.42 μM. | 30245402 | |
| HEK293 | Cytotoxicity assay | 48 hrs | Cytotoxicity against HEK293 cells after 48 hrs by resazurin dye based assay, IC50 = 1.42 μM. | 30245402 | |
| RAW264.7 | Anti-inflammatory assay | 1 hr | Anti-inflammatory activity in LPS-stimulated mouse RAW264.7 cells assessed as suppression of nitric oxide production pre-incubated for 1 hr before LPS stimulation for 24 hrs by Griess reagent based assay, IC50 = 2.2 μM. | 25113875 | |
| RAW264.7 | Anti-inflammatory assay | 1 hr | Anti-inflammatory activity in LPS-stimulated mouse RAW264.7 cells assessed as suppression of TNFalpha production pre-incubated for 1 hr before LPS stimulation for 24 hrs by ELISA method, IC50 = 4.7 μM. | 25113875 | |
| RAW264.7 | Anti-inflammatory assay | 1 hr | Anti-inflammatory activity in LPS-stimulated mouse RAW264.7 cells assessed as suppression of PGE2 production pre-incubated for 1 hr before LPS stimulation for 24 hrs by enzyme immunoassay method, IC50 = 8.28 μM. | 25113875 | |
| HEK293 | Function assay | Inhibition of HDAC6 in HEK293 cells, IC50 = 0.015 μM. | 18308563 | ||
| HEK293 | Function assay | Inhibition of HDAC1 in HEK293 cells, IC50 = 0.018 μM. | 18308563 | ||
| HeLa | Function assay | Inhibition of HDAC in human HeLa cells using Fluor de Lys as substrate by fluorescence assay, IC50 = 0.027 μM. | 23639537 | ||
| HeLa | Function assay | Inhibition of HDAC from human HeLa cells, IC50 = 0.028 μM. | 18247554 | ||
| HEK293 | Function assay | Inhibition of HDAC3 in HEK293 cells, IC50 = 0.046 μM. | 18308563 | ||
| HCT116 | Antiproliferative assay | Antiproliferative activity against human HCT116 cells assessed as growth inhibition, IC50 = 0.16 μM. | 21650221 | ||
| HCT116 | Antiproliferative assay | Antiproliferative activity against human HCT116 cells, IC50 = 0.16 μM. | 21742496 | ||
| H1299 | Antiproliferative assay | Antiproliferative activity against human H1299 cells, IC50 = 0.46 μM. | 21650221 | ||
| 他の多くの細胞株試験データをご覧になる場合はこちらをクリックして下さい | |||||
生物活性
| 製品説明 | Belinostatは、細胞フリーアッセイで27 nMのIC50を持つ新規のHDAC阻害剤であり、シスプラチン耐性腫瘍で活性が実証されています。Belinostat(PXD101)はautophagyを誘導します。 | ||
|---|---|---|---|
| 特性 | Lead compound of Topotarget. | ||
| Targets |
|
| In Vitro | ||||
| In vitro | Belinostat inhibits the growth of tumor cells (A2780, HCT116, HT29, WIL, CALU-3, MCF7, PC3 and HS852) with IC50 from 0.2-0.66 μM. PD101 shows low activity in A2780/cp70 and 2780AD cells, which are cisplatin and doxorubicin-resistant derivatives of A2780 cells. This compound could induce apoptosis through PARP cleavage and acetylation of histones H3/H4. It inhibits bladder cancer cell growth, especially in 5637 cells, which shows accumulation of G0-G1 phase, decrease in S phase and increase in G2-M phase. The growth inhibitory activity of this chemical on cell lines is not strongly influenced by the multidrug-resistant phenotype, whereas the activity of docetaxel is clearly affected. It could enhance the growth inhibitory activity of docetaxel or carboplatin in OVCAR-3 and A2780 cells. This compound also shows enhanced tubulin acetylation in ovarian cancer cell lines. A recent study shows that it activates protein kinase A in a TGF-β signaling-dependent mechanism and decreases survivin mRNA. | |||
|---|---|---|---|---|
| Kinase Assay | Histone Deacetylase Activity | |||
| Subconfluent cultures are harvested and washed twice in ice cold PBS and pelleted by centrifugation at 200 × g for 5 min. The cell pellet is resuspended in two volumes of lysis buffer [60 mM Tris buffer (pH 7.4) containing 30% glycerol and 450 mM NaCl] and lysed by three freeze (dry ice) thaw (30 °C water bath) cycles. Cell debris is removed by centrifugation at 1.2 × 104 g for 5 min, and the supernatant is stored at −80 °C. Histone H4 peptide (sequence SGRGKGGKGLGKGGAKRHRK corresponding to the 20 NH2-terminal residues) is acetylated by a recombinant protein containing the hypoxanthine-aminopterin-thymidine domain of p300, using [3H]acetyl CoA as a source of acetate. H4 peptide (100 μg) is mixed with hypoxanthine-aminopterin-thymidine buffer (50 mM Tris HCl pH 8.0, 5% glycerol, 50 mM KCl, and 0.1 mM EDTA), 1 mM DTT, 1 mM 4-(2-aminoethyl) benzenesulfonylfluoride, 1 × complete protease inhibitors, 50 μL of purified p300, and 1.85 m [3H]acetyl CoA (4.50Ci/mmol) in a final volume of 300 μL and incubated at 30 °C for 45 min. The p300 protein is removed by incubation with 20 μL of 50% Ni-agaroase beads for 1 hour at 4 °C and centrifugation. The supernatant is applied to a 2 mL Sephadex G15 column, and the flow through is collected. One milliliter of distilled H2O is gently applied, and three drop fractions are collected; this is repeated until 4–5 mL of distilled H2O has been added, and ∼40 fractions are collected. Three microliters of each fraction are diluted in 2 mL of scintillation fluid and counted in a scintillation counter to identify the fractions containing the labeled peptide. These fractions are pooled, and 1 μL of the combined sample is measured to assess the radioactivity in every peptide batch (3-7×103 cpm/μL). For activity assays, the reaction is carried out in a total volume of 150 μL of buffer [60 mM Tris (pH 7.4) containing 30% glycerol] containing 2 μL of cell extract and, where used, 2 μL of this compound. The reaction is started by the addition of 2 μL of [3H] labeled substrate (acetylated histone H4 peptide corresponding to the 20 NH2-terminal residues). Samples are incubated at 37 °C for 45 min, and the reaction stopped by the addition of HCl and acetic acid (0.72 and 0.12 M final concentrations, respectively). Released [3H]acetate is extracted into 750 μL of ethyl acetate, and samples are centrifuged at 1.2× 104 g for 5 min. The upper phase (600 μL) is transferred to 3 mL of scintillation fluid and counted. | ||||
| 細胞実験 | 細胞株 | A2780, A2780/cp70, 2780AD, HCT116, HT29, WIL, CALU-3, MCF7, PC3 and HS852 | ||
| 濃度 | 0.016 - 10 μM | |||
| 反応時間 | 24 hours | |||
| 実験の流れ | Tumor cell lines are seeded in 5 mL of medium at a density of 8 × 104 cells/25 cm2 flask and incubated for 48 hours. Cells are exposed to Belinostat (0.016 to 10 μM) for 24 hours. The medium is removed, and 1 mL of trypsin/EDTA is added to each flask. Once the cells have detached, 1 mL of medium is added, the cells are resuspended, and those from the control untreated flask are counted. Cells are diluted and plated into 6-cm Petri dishes (three per flask) at a density of 0.5-2× 103 cells/dish depending on the cell line. Cells from the drug-treated flasks are diluted and plated as for the control flasks. Dishes are incubated for 10–15 days at 37 °C. Cells are washed with PBS, fixed in methanol, and stained with crystal violet, and colonies that contained ≥50 cells counted. Sensitivity is expressed as the IC50 defined as the concentration of this compound required to reduce the number of colonies to 50% of that of the control untreated cells. | |||
| 実験結果図 | Methods | Biomarkers | 結果図 | PMID |
| Western blot | p-H2AX(Ser139) / KU70 / KU80 / RAD51 / RAD52 / ERCC1 Acetyl Histone H3 / Acetyl Histone H4 / Acetyl tubulin p21 / p27 SOS1 / SOS2 PARP / p-ERK / p-p38 / p38 / p-BRAF / p-MEK / MEK |

|
24155971 | |
| Growth inhibition assay | Cell viability IC50 |
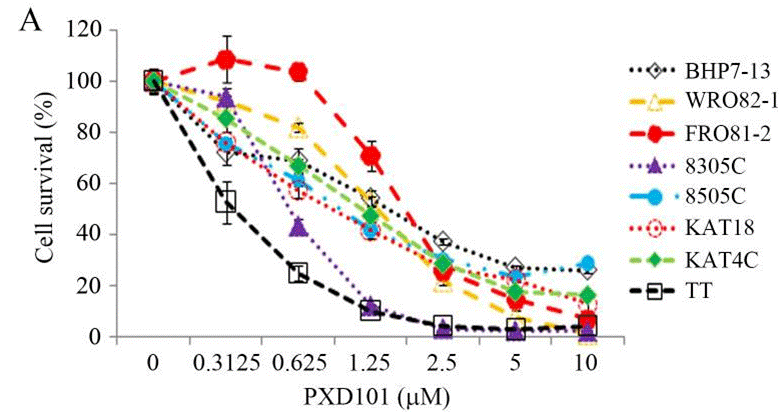
|
24155971 | |
| In Vivo | ||
| In Vivo | Belinostat indicates significant tumor growth delay in A2780 and A2780/cp70 xenograft at a dose of 10mg/kg with no effects on the body weight. This compound also induces p21WAF1, HDAC core and cell communication genes in mouse bladder tumors. Its monotherapy induces dose-proportional antitumor effects with TGI of 47% at a dose of 100mg/kg in A2780 xenograft. The combination of this chemical (100 mg/kg) with carboplatin (40 mg/kg) could delay tumor growth from 18.6 days to 22.5 days. Combining with bortezomib, it results in great tumor inhibition and gastrointestinal toxicity in mice with bortezomib-resistant UMSCC-11A xenograft. | |
|---|---|---|
| 動物実験 | 動物モデル | A2780, A2780/cp70 and HCT116 cells are injected s.c. into the right flank of CD1 nu/nu mice. |
| 投与量 | ≤40 mg/kg | |
| 投与経路 | Administered via i.p. | |
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT06406465 | Not yet recruiting | Carcinoma Neuroendocrine|Tumor Neuroendocrine|Tumors Neuroendocrine|Neuroendocrine; Carcinoma|Small Cell; Receptors |
National Cancer Institute (NCI)|National Institutes of Health Clinical Center (CC) |
May 15 2024 | Phase 2 |
| NCT04315233 | Recruiting | Metastatic Breast Cancer|Recurrent Ovarian Carcinoma |
University of Utah|Novartis|Acrotech Biopharma |
May 3 2021 | Phase 1 |
| NCT04703920 | Recruiting | Metastatic Breast Cancer|Metastatic Castration-resistant Prostate Cancer|Metastatic Ovarian Carcinoma |
University of Michigan Rogel Cancer Center|Pfizer|Acrotech Biopharma Inc. |
March 4 2021 | Phase 1 |
| NCT03772925 | Active not recruiting | Recurrent Acute Myeloid Leukemia|Recurrent Myelodysplastic Syndrome|Refractory Acute Myeloid Leukemia|Refractory Myelodysplastic Syndrome |
National Cancer Institute (NCI) |
June 20 2019 | Phase 1 |
|
化学情報
| 分子量 | 318.35 | 化学式 | C15H14N2O4S |
| CAS No. | 866323-14-0 | SDF | Download Belinostat (PXD101) SDFをダウンロードする |
| Smiles | C1=CC=C(C=C1)NS(=O)(=O)C2=CC=CC(=C2)C=CC(=O)NO | ||
| 保管 | |||
|
In vitro |
DMSO : 64 mg/mL ( (201.03 mM); 吸湿したDMSOは溶解度を減少させます。新しいDMSOをご使用ください。) Water : Insoluble Ethanol : Insoluble |
モル濃度計算器 |
|
in vivo Add solvents to the product individually and in order. |
投与溶液組成計算機 | |||||
実験計算
投与溶液組成計算機(クリア溶液)
ステップ1:実験データを入力してください。(実験操作によるロスを考慮し、動物数を1匹分多くして計算・調製することを推奨します)
mg/kg
g
μL
匹
ステップ2:投与溶媒の組成を入力してください。(ロット毎に適した溶解組成が異なる場合があります。詳細については弊社までお問い合わせください)
% DMSO
%
% Tween 80
% ddH2O
%DMSO
%
計算結果:
投与溶媒濃度: mg/ml;
DMSOストック溶液調製方法: mg 試薬を μL DMSOに溶解する(濃度 mg/mL, 注:濃度が当該ロットのDMSO溶解度を超える場合はご連絡ください。 )
投与溶媒調製方法:Take μL DMSOストック溶液に μL PEG300,を加え、完全溶解後μL Tween 80,を加えて完全溶解させた後 μL ddH2O,を加え完全に溶解させます。
投与溶媒調製方法:μL DMSOストック溶液に μL Corn oil,を加え、完全溶解。
注意:1.ストック溶液に沈殿、混濁などがないことをご確認ください;
2.順番通りに溶剤を加えてください。次のステップに進む前に溶液に沈殿、混濁などがないことを確認してから加えてください。ボルテックス、ソニケーション、水浴加熱など物理的な方法で溶解を早めることは可能です。
技術サポート
ストックの作り方、阻害剤の保管方法、細胞実験や動物実験の際に注意すべき点など、製品を取扱う時に問い合わせが多かった質問に対しては取扱説明書でお答えしています。
他に質問がある場合は、お気軽にお問い合わせください。
* 必須
よくある質問(FAQ)
質問1:
Could you please give some suggestions for the use of it in vivo (i.p. injection)?
回答
For I.P. injection, it can be dissolved in 2% DMSO+30% PEG 300+ddH2O at 10 mg/ml clearly. When preparing the solution, please dissolve this compound in DMSO clearly first. Then add PEG, after they mixed well, then dilute with water. Hope this information is useful to you.